
Solid objects, such as your desk or a rod of aluminum, can conduct heat. The magnitude of the thermal diffusivity of the material determines how quickly the heat moves through a given amount of material. The equation for thermal diffusivity (a) is given by:
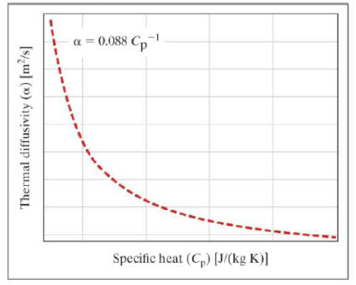 Experiments are conducted to change the specific heat (Cp) of the material while holding the thermal conductivity (k) and the density (ρ) constant. The results are shown graphically.
Experiments are conducted to change the specific heat (Cp) of the material while holding the thermal conductivity (k) and the density (ρ) constant. The results are shown graphically.
- a. What are the units of the constant 0.088? Simplify your answer.
- b. If the thermal conductivity of the material is 237 watts per meter kelvin, what is the density of the material?
- c. If the material has a density of 4500 kilograms per cubic meter, what is the thermal conductivity of the material in units of watts per meter kelvin?
Learn your wayIncludes step-by-step video

Chapter 12 Solutions
Thinking Like an Engineer: An Active Learning Approach (4th Edition)
Additional Engineering Textbook Solutions
Engineering Mechanics: Statics
Automotive Technology: Principles, Diagnosis, And Service (6th Edition) (halderman Automotive Series)
Engineering Mechanics: Dynamics (14th Edition)
Automotive Technology: Principles, Diagnosis, and Service (5th Edition)
Statics and Mechanics of Materials (5th Edition)
- 1.77 Explain each in your own words. (a) What is the mode of heat transfer through a large steel plate that has its surfaces at specified temperatures? (b) What are the modes when the temperature on one surface of the steel plate is not specified, but the surface is exposed to a fluid at a specified temperature?arrow_forwardThe following graph shows the thermal behavior of 2 kg of a material called Uniandesato undergoing a solid-liquid phase transition. In a container, thermally insulated from the outside, 20 kg of liquid water at a temperature of 80°C are placed. In addition to this, an unknown amount of Uniandesato in a 100% solid state at its melting temperature (10°C) is added. The specific heat of water is 4186 J/kg°C. a) If the system reaches an equilibrium temperature of 60°C, calculate the initial amount of Uniandesato added to the container. b) Calculate the change in entropy during this process and show that it is consistent with the Second Law of Thermodynamics. Hint: Extract the necessary information to solve this problem from the graph.arrow_forwardWhen the sun is high in the sky, it delivers approximately 1000 watts of power to each square meter of earth's surface. The temperature of the surface of the sun is about 6000 K, while that of the earth is about 300 K. Suppose you plant grass on this square meter of earth. Some people might argue that the growth of the grass (or of any other living thing) violates the second law of thermodynamics, because disorderly nutrients are converted into an orderly life form. How would you respond?arrow_forward
- Which of the following statements are correct in the context of thermal conductivity? (Check all that apply.) Check All That Apply The thermal conductivity of gases is proportional to the square root of absolute temperature. The thermal conductivity of liquids is proportional to the square root of absolute temperature. The thermal conductivity of most liquids decreases with increasing temperature. The thermal conductivity of most liquids increases with increasing temperature.arrow_forwardAn electric heater producing 260 W of heat is used to warm up a room containing 7 m3 of air. If we assume the room is perfectly sealed and there is no heat loss through the room boundaries, such that all of the heater output goes into increasing the air temperature, how long will it take to heat up the air in the room from 5.0 °C to 24.1 °C? Give your answer to the nearest minute and assume that the specific volume (v = 0.85 m3/kg) and specific heat capacity at constant volume (cv = 1.005 kJ/(kg K)) remain constant throughout the heating process.arrow_forwardA solid steel cube is stored in a freezer, where the temperature is −20.0 ̊C. While the cube is in the freezer, you measure its side length (L) to be 1.75050 meters. The cube is then placed outside for a long time, so that the temperature of the cube reaches the temperature of the air outside. As a result, the side length increases to 1.75170 meters due to thermal expansion. The coefficient of linear expansion for steel is 12.0×10−6 ̊C−1. (a) What is the temperature of the air outside? Give your answer in degrees Celsius ( ̊C). (b) What is the volume of the cube when its temperature is 20.0 ̊C? Give your answer in cubic meters (m3).arrow_forward
- Shape Factor Conduction Problem A cylindrical pipeline that is used for the transport of crude oil is buried in the soil horizontally such that its centerline is 1.5 m (z) below the surface. The pipe has the outer diameter of 0.5 m (D) and is coated with a 100 mm thick layer of glass insulation on the outside. Assume that heated oil at 120 °C flows through the pipe and the soil surface temperature is at 0 °C (T2). The soil thermal conductivity is known as 0.5 W/m-K, and the glass insulation thermal conductivity is known as 0.07 W/m-K. What is the rate of heat loss per unit length of the pipe (W/m)? Soil Glass insulation Oil, Tarrow_forwardA temperature difference of 85 •C is impressed across a fiberglass layer of 13 cm thickness. The thermal conductivity of the fiberglass is 0.035 W/m • -C. Compute the heat transferred through the material in kcal per hour per unit square meter area.arrow_forward8) Consider two liquids, A and B. with temperatures Te > TA. The two objects are put into thermal contact for a time period. Without just saying 'heat flows from hot to cold' how would you prove to someone that a quantity of heat flowed from B to Á. (think of James Joule's experiments) 9) If the temperature of the sun were to suddenly double, by what multiplicative factor would the thermal radiation change ? Show Workarrow_forward
- . A thermocouple is a temperature-measurement device that consists of two dissimilar metal wires joined at one end. An oversimplified diagram follows. Metal 1 ΡOTENTIOMΕΤER V(mV) Metal 2 A voltage generated at the metal junction is read on a potentiometer or millivoltmeter. When certain metals are used, the voltage varies linearly with the temperature at the junction of the two metals: V(mV) = aT(°C) +b An iron-constantan thermocouple (constantan is an alloy of copper and nickel) is calibrated by inserting its junction in boiling water and measuring a voltage V = 5.27 mV, and then inserting the junction in silver chloride at its melting point and measuring V = 24.88 mV. (a) Derive the linearequation for V(mV) in terms of T(°C). Then convert it to an equation for T in terms of V. (b) If the thermocouple is mounted in a chemical reactor and the voltage is observed to go from 10.0 mV to 13.6 mV in 20 s, what is the average value of the rate of change of temperature, dTldt, during the…arrow_forward4. For a particular thermocouple, if one junction is maintained at 0°C (cold junction) and the other junction is used as a probe to measure the desired Celsius temperature t, the voltage V generated in the circuit related to the temperature t as V = t (a + bt) Further, for this thermocouple, when V is in millivolts, the two constants are a = 0.25 and b = -5.5 x10+. Determine the value of V if the measured temperature is 100°C.arrow_forwardProblem 1. 67 pts bar shown below, determine the temperatures at Nodes 2 and 3. Assume 1-D heat transfer that only occurs in the x-direction as the upper and lower 1-D Heat Transfer with Conduction. For the 1-D composite boundaries of the elements are insulated. Assume the cross-sectional area is the same for all elements, A=0.01 m?. For Element 1, let the thermal conductivity be 100 W/(m °C). For Element 2, let the thermal conductivity be 110 W/(m °C). For Element 3, let the thermal conductivity be 120 W/(m °C). The left end of the bar has a constant temperature of 120 °C (at Node 1) and the right end has a constant temperature of 276 °C (at Node 4). Insulated, 1-d heat transfer in x-dir Node 1 Node 2 Node 3 Node 4 +x 120°C E1 E2 ЕЗ 276°C 1 mm 2 mm 0.5mm Insulatedarrow_forward
 Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
