
Concept explainers
a.
Interpretation:
For the below compound the chirality centers has to be labeled and number of chiral centers should be determined
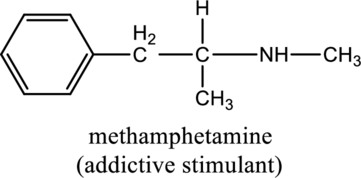
Concept introduction:
Chirality center:
When a carbon atom is bonded to four different groups, then that carbon is named as chiral carbon and it is called as chirality center. Generally, the carbon atom contains tetrahedral valency, therefore it bonded to four atoms or molecules. The chiral center is bonded with four different elements or groups. If the single carbon atom is bonded to two or more identical group or atom, then it is also not considered as chiral carbon. In addition to this, if the carbon atom contains multiple bond then it does not able to bond with four different groups then it is not considered to be as a chirality center.
b.
Interpretation:
For the below compound the chirality centers has to be labeled and number of chiral centers should be determined
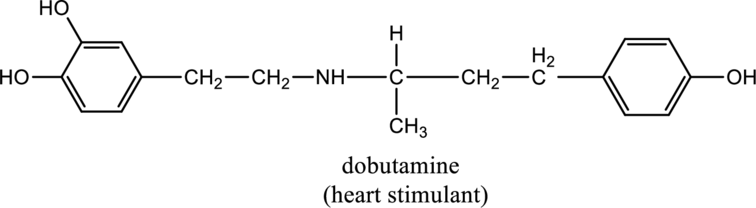
Concept introduction:
Refer to part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Principles of General, Organic, Biological Chemistry
- 18-18 Propanoic acid and methyl acetate are constitutional isomers, and both are liquids at room temperature. One of these compounds has a boiling point of 141°C; the other has a boiling point of 57°C. Which compound has which boiling point? Explain.arrow_forwardReactions (chemical equation) of test samples with Tollen's reagent: Cyclohexanone Acetone Benzaldehyde Glucose Fructosearrow_forwardWhich of the following sweet molecule and its classification is correct? cyclamate: artificial carbohydrate sweetener phylloducin:artificial nutritive sweetener fructose: natural non-nutritive sweetener maltitol:reduced carbohydrate nutritive sweetenerarrow_forward
- How many kinds of chemically non-equivalent carbons are there in each of the following compounds? a b علم CH₂-CH₂- +NH₂ OH CH₂ Aspirin (analgesic, antipyretic and antibiotic) The number of chemically non-equivalent carbons is The number of chemically non-equivalent carbons isarrow_forwardGive an acceptable name for each compound depicted in the ball-and-stick models.arrow_forwardCompound X and compound Y are constitutional isomers with the molecular formula C5H10. Compound X possesses a carbon-carbon double bond in the trans configuration, while compound Y possesses a carbon-carbon double bond that is not stereoisomeric: Draw the structure of compound X. Draw Your Solutionarrow_forward
- Consider the following two molecules and answer the following questions. Molecule A: HO- CH-CH3 CH3 Molecule B: || H,C-CH,-C-0-CH3 a) What are all of the IMFS present in molecule A? b) What are all of the IMFS present in molecule B? c) Which would have the higher boiling point, molecule A or molecule B? d) What is the relationship between molecules A & B: structural isomers, geometric isomers, identical, or not related?arrow_forwardReactions of test samples with Tollen's reagent: Cyclohexanone Acetone Benzaldehyde Glucose Fructosearrow_forwardIdentify the functional groups in two drugs, atenolol and donepezil. Atenololis a β (beta) blocker, a drug used to treat hypertension (high bloodpressure), and donepezil (trade name Aricept) is used to treat mild tomoderate dementia associated with Alzheimer's disease.arrow_forward
- CH3CH2OH CH3SH CH3CH2CH3 Which compound can form hydrogen bonds? Which compound has the highest boiling point? Which compound has the lowest boiling point?arrow_forwardList the compounds in order of decreasing boiling point. CH3CH2CH3 He CH3CHOarrow_forwardBile salts are derivative of cholestrol. However, the solubilities of these compounds in water are drastically different; cholestrol is highly hydrophobic, and the bile salts are soluble in digestive juices. Explain the differences.arrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



