
Concept explainers
(a)
Interpretation:
The product obtained by the reaction of the given
Concept Introduction:
Hydrogenation reaction is nothing but the addition of hydrogen to an alkene. In this reaction two bonds are simultaneously broken, one is carbon-carbon double bond, and second one is hydrogen-hydrogen bond, finally forms two carbon-hydrogen bonds. The general reaction can be represented as follows,
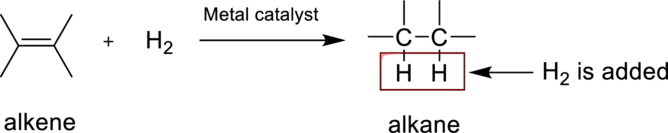
(b)
Interpretation:
The product obtained by the reaction of the given alkene with hydrogen and palladium catalyst has to be identified.
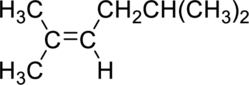
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The product obtained by the reaction of the given alkene with hydrogen and palladium catalyst has to be identified.
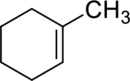
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Principles of General, Organic, Biological Chemistry
- Draw the products of combustion of each alkane. a. CH;CH,CH,CH2CH(CH3)2 b.arrow_forward8. Give the IUPAC name for each alkene. CH3 CH2CH,CH3 Br а.arrow_forwardDraw the products formed when CH3CH2C ≡ C−Na+reacts with each compound. a. CH3CH2CH2Brb. (CH3)2CHCH2CH2Clc. (CH3CH2)3CCld. BrCH2CH2CH2CH2OHe. ethylene oxide followed by H2Of. propene oxide followed by H2Oarrow_forward
- 3. What alcohol is formed when each alkene is treated with H2O in the presence of H2SO4 (as the catalyst)? а. СНЗСН-СНСНЗ b. CH3CH2CH=CH2 С. CH3 CH3arrow_forwardPLEASE HELP Draw the products formed when cyclohexene is treated with each reagent.arrow_forwardDoes the equilibrium favor the reactants or products in each substitution reaction? a. CH;CH2-NH2 Br CH;CH2-Br + "NH2 b. "CN CN + I-arrow_forward
- 14. Choose the reagents needed to create an alkene with a double bond between C3 and C4. a. Conc. H2SO4 OH b. 1) BH3 THF; 2) H2O2, NaOH c. HBr, ROOR d. HBrarrow_forwardDraw the products formed when each alkene is treated with HCI. CH3 CH3 b. c=CH2 a. С. CH3arrow_forwardGive the IUPAC name for each compound. CH3 CH2CH3 Br a. PHCH(CH3)2 b. С. d.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





