
(a)
Interpretation:
The balanced equation to represent the complete combustion of the given compound is to be stated.
Concept introduction:
Alkane is made of only carbon and hydrogen atoms. These atoms form a bond with each other and with other atoms through the sharing of electrons. They do not completely donate their electrons to other atoms while forming a bond. A general complete combustion reaction of
Answer to Problem 11.60E
The balanced equation to represent the complete combustion of the given compound is shown below as,
Explanation of Solution
The given molecule is butane. The chemical formula of butane is
A general complete combustion reaction of alkanes is shown below as,
Where,
•
The number of carbon atoms in butane is four.
Therefore, the balanced equation to represent the complete combustion of the given compound is shown below as,
The reaction is further simplified as,
The balanced equation to represent the complete combustion of the given compound is shown below as,
(b)
Interpretation:
The balanced equation to represent the complete combustion of the given compound is to be stated.
Concept introduction:
Alkane is made of only carbon and hydrogen atoms. These atoms form a bond with each other and with other atoms through the sharing of electrons. They do not completely donate their electrons to other atoms while forming a bond. A general complete combustion reaction of alkanes is shown below as,
Answer to Problem 11.60E
The balanced equation to represent the complete combustion of the given compound is shown below as,
Explanation of Solution
The given molecule is shown below as,
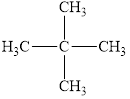
Figure 1
The chemical formula of the given compound is
A general complete combustion reaction of alkanes is shown below as,
Where,
•
The number of carbon atoms in the given compound is five.
Therefore, the balanced equation to represent the complete combustion of the given compound is shown below as,
The balanced equation to represent the complete combustion of the given compound is shown below as,
(c)
Interpretation:
The balanced equation to represent the complete combustion of the given compound is to be stated.
Concept introduction:
Alkane is made of only carbon and hydrogen atoms. These atoms form a bond with each other and with other atoms through the sharing of electrons. They do not completely donate their electrons to other atoms while forming a bond. A general complete combustion reaction of cycloalkanes is shown below as,
Answer to Problem 11.60E
The balanced equation to represent the complete combustion of the given compound is shown below as,
Explanation of Solution
The given molecule is shown below as,
![]()
Figure 2
The chemical formula of the given compound is
A general complete combustion reaction of cycloalkanes is shown below as,
Where,
•
The number of carbon atoms in the given compound is four.
Therefore, the balanced equation to represent the complete combustion of the given compound is shown below as,
The balanced equation to represent the complete combustion of the given compound is shown below as,
Want to see more full solutions like this?
Chapter 11 Solutions
Chemistry for Today: General, Organic, and Biochemistry
- Consider the following isomers of hexene: A. a disubstituted cis- alkene B. the geometric isomer of ISO C. a tri-substituted Z- alkene D. most stable alkene E. has the highest boiling point F.arrow_forwardHow many water molecules would be produced by the complete combustion of one straight chained butane molecule? A. 4 B. 5 C. 6 D. 8arrow_forwardWrite the chemical equations for tollen's test reaction for (Use structural formula) a. n-hexane b. cyclohexene c. benzene d. toluenearrow_forward
- Draw all possible the structural formula and give the IUPAC name of the following compounds:a. trinitrophenol b. dichlorobenzoic acid c. bromoanthracenearrow_forwardDraw line formulas for each of the following alcohols: a. sec-Hexyl alcohol b. Isopentyl alcohol c. Cyclobutyl alcohol d. Hexyl alcoholarrow_forward1. Identify the IUPAC name for the following alkane: a. C. Butane Pentane b. Propane d. Crownanearrow_forward
- A primary alcohol has a hydroxyl group bonded to a(n) a. aromatic carbon. b. trisubstituted carbon. c. disubstituted carbon. d. singly substituted or unsubstituted carbon.arrow_forwardName the following compounds a. b. c.arrow_forward12. Give IUPAC names for the following compounds- A. B. C. OH ethylpropamatearrow_forward
- 7. The reaction of water with an alkene to produce alcohol is a/an ______ reaction.A. Addition B. Condensation C. Combustion D. Saponificationarrow_forwardAlkenes are not soluble in water because they are a. polar b. hydrophilic c. hydrophobic d. less dense than waterarrow_forwardDraw the following compounds and give it's IUPAC name: a. Propyl mercaptan b. Isobutyl mercaptan c. 1-Butanethiol d. 2-Propanethiolarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





