
(a)
Interpretation:The mechanism for the formation of molecule 26 from molecule 28 through a hydride shift followed by deprotonation needs to be explained.
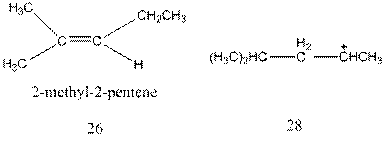
Concept Introduction : A carbocation rearrangement in which a hydrogen atom migrates from the carbon to the carbon possessing formal charge of one from the adjacent carbonis called hydride shift.
(b)
Interpretation:The mechanism for formation of 27 from 28 needs to be determined.
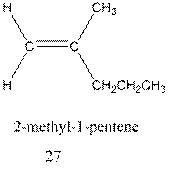
Concept Introduction : A carbocation rearrangement in which a hydrogen atom migrates from the carbon to the carbon possessing formal charge of one from the adjacent carbonis called hydride shift.
(c)
Interpretation:The mechanism of formation of 26 through acid catalysed rearrangement of either isomer of 4-methyl-2-pentene and formation of 27 by addition of a proton to 26 followed by deprotonation of intermediate carbocation needs to be explained.
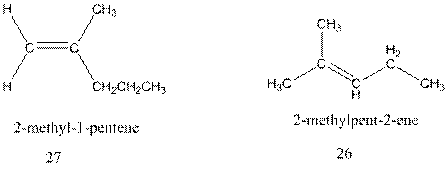
Concept Introduction : A carbocation rearrangement in which a hydrogen atom migrates from the carbon to the carbon possessing formal charge of one from the adjacent carbonis called hydride shift.
(d)
Interpretation:The different in the two types of mechanism for the formation of 26 and 27 needs to be explained.
Concept Introduction:Mechanism of a
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- Draw the structure of the major product of following reactions: CH2l2, Zn(Cu) Et,0 „MgBr H3O* to H,0 2. Zn 3. ..... HCI CH212, Zn(Cu) 4. Et,0 H3O* + CH;CH2M9B. 5,arrow_forward2. Name and Draw the structures p-chloroaniline of all possible chemical (Electrophilic aronmatic substitution) reactions of the compound namely a. Halogenation (Chlorination or Bromination) b. Nitration c. Sulphonation d. Friedal Craft Alkylation e. Eriedal Craft Acylationarrow_forward26) What type of reaction is depicted below? NaCe thenH* BO a. 1,2-addition b. acyl substitution c. conjugate addition d. SN reaction e. an impossible one; carbanions do not add to alkenesarrow_forward
- 46. Which carbonyl group of the given compound is most reactive for nucleophilic addition reaction? A. Carbonyl Group 1 B. Carbonyl Group 2 C.Carbonyl Group 3 D. All have equal reactivity 47. Which of the following does NOT give a brick red precipitate with Fehling solution? A Acetone B. Acetaldehyde C. Formalin D. D-glucose 48. Which of the following structures contains a hemiacetal group CO OH H₂C 0 OCH, A OH OCH H₂C-C-CH₂ C C.H. CH OCH OCH, barrow_forward37. Under acidic condition, which of the following compounds can not be oxidized by KMnO4. A. toluene B. 2-butene C. methylcyclohexane D. 2-butyne 38. What alkene product can be produced from the reaction of 2-hexyne with Lindlar's catalyst? A. cis-2-Hexene B. trans-2-Hexene C. Hexane D. A and Barrow_forwardShow the reaction mechanism for the formation of a. RCONH2 from the reaction of RCOOR' with NH3 b. RCOOR" from the reaction of RCOOR' with R"OH in acidic mediumarrow_forward
- 37- The Sulphonation of benzene ring proceeds by using . ???? SO,H (A) Conc.HNO3 and conc. H2S04 (C) Conc. H2SO4 (B) Dil. H2SO4 (D) SO3/ H2SO4 38- Which of the following benzene derivative is the most reactive and ortho, para director? CHO NH. (A) (B) (C) (D) 39- What is the major product for the following reaction? NO, HNO, ???? H,SO, NO, NO, NO, NO (A) NO, (B) NO, (D) None of these 40- a – D - glucose differ from B-D-glucose (A) Because they are anomer (B) Because they are enantiomers (C) Because they are mirror image of each other (D) Because they are geometrical isomers 41-Ninhydrin test is given by (C) Proteins (D) Carbohydrates (B) Alcohols (A) Alkynes 42- Which test of the following can distinguish between 1°, 2° and 3° amines : (C) Acidic Test (D) None (B) Tolen's Test (A) The Hinsberg Test 43- How many a – amino acid are there in animal protein : (D) 10 (B) 20 (C) 15 (A) 55arrow_forwardi. LIAIH4 i. deprotection J ii. dehydration .CO2Me CH2=CHCOCH3 protection ii. H30* K The synthesis of K from H involves a 3-steps reaction as shown above. Hydrolysis of H followed by heating produces compound L. Give IUPAC name for L. Write the reaction mechanism involved during the heating process that brought iii. to the formation of L.arrow_forward15. Give the major organic product, paying attention to stereochemistry (syn/anti). Indicate if a racemic mixture is expected. a. a. BH3 b. H2O2, OH a. BH3 b. b. H2O2, OH a. BH3 a. BH3 C. d. b. H2O2, OH b. H₂O2, OH e. a. BH3 b. H₂O2, OHarrow_forward
- a. Use the following reaction to determine how much of each product would be releasedif 42 000 kg (42 tonnes) of methyl isocyanate reacted with excess water:methyl isocyanate + water → 1,3-dimethyl urea + carbon dioxide2C2H3NO + H2O → C3H8N2O + CO2arrow_forwardselect reagents: cyclopentanone Reagents a. C6H5CHO b. NaOH, ethanol c. Pyrrolidine, cat. Hj. d. H₂C=CHCN e. H3O+ f. LDA g. A) bg EtOC(=O)CO₂Et h. i. B) ka k. I. m. O: BrCH₂CH=CH₂ NatOEt, ethanol Br₂, H+ K+ t-BUO™ CH2(CO2Et)z heat C) jh CO₂Et D) Imarrow_forwardbläi 5 Q3: 5. Select the product of the .following reaction 1. Mg 2. co, followed by acid workup 3. Pyridine NICH)2 он NICH2 NCH2 NICH)2 A. В. C. D.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





