
Human Anatomy & Physiology (11th Edition)
11th Edition
ISBN: 9780134580999
Author: Elaine N. Marieb, Katja N. Hoehn
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
(cell biology/human physiology)
1) Is it true that AMP and ADP levels are similarlyly indicators of energy status (as shown by ATP concentration)
2) Explain the change of peaks from I to V in the sequence of events from A to D?
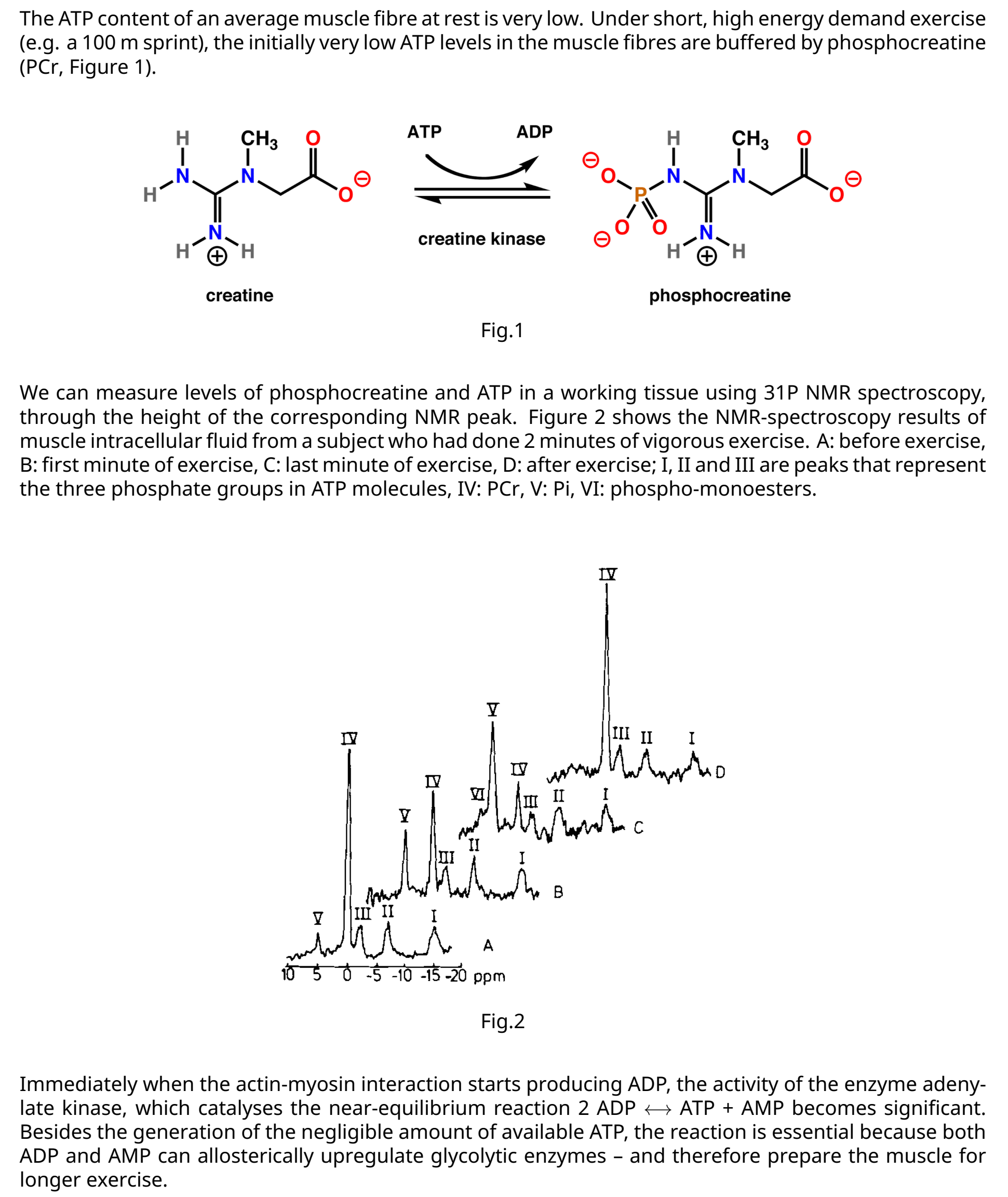
Transcribed Image Text:The ATP content of an average muscle fibre at rest is very low. Under short, high energy demand exercise
(e.g. a 100 m sprint), the initially very low ATP levels in the muscle fibres are buffered by phosphocreatine
(PCr, Figure 1).
ATP
ADP
Н
CHз
Н
CHз
.N.
N.
'N'
'N'
.N.
creatine kinase
H'
Н
H.
creatine
phosphocreatine
Fig.1
We can measure levels of phosphocreatine and ATP in a working tissue using 31P NMR spectroscopy,
through the height of the corresponding NMR peak. Figure 2 shows the NMR-spectroscopy results of
muscle intracellular fluid from a subject who had done 2 minutes of vigorous exercise. A: before exercise,
B: first minute of exercise, C: last minute of exercise, D: after exercise; I, II and III are peaks that represent
the three phosphate groups in ATP molecules, IV: PCr, V: Pi, VI: phospho-monoesters.
IV
III II
II
III' II
o $ -10 -15 -20
ppm
Fig.2
Immediately when the actin-myosin interaction starts producing ADP, the activity of the enzyme adeny-
late kinase, which catalyses the near-equilibrium reaction 2 ADP + ATP + AMP becomes significant.
Besides the generation of the negligible amount of available ATP, the reaction is essential because both
ADP and AMP can allosterically upregulate glycolytic enzymes - and therefore prepare the muscle for
longer exercise.
I.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Based on the pathways of the effects of supplementing α-linolenic acid (ALA) and docosahexaenoic acid (DHA) on the composition of long-chain fatty acids in cell membranes, discuss the mechanism further in the consumption of eicosapenpentaenoic acid (EPA) for heart protection.arrow_forwardDigoxin, a toxin derived from the foxglove (shown), can be used to treat heart disorders such as atrial fibrillation. Digoxin’s mechanism of action is to inhibit the Na+/K+ ATPase. Which of the following is the most likely side effect of Digoxin treatment? Failure to transport glucose into cells during an extended fast Failure to transport glucose into cells after after eating a large meal Enhanced response of muscle cells to Acetylcholine Failure to transport Cl- out of the cell using direct active transportarrow_forwardProtein Phosphatase 1: What overall effect does PP1 have on the storage or breakdown of glycogen? (1 point) How does PPI regulate the activity of its targets? (1 point) Which enzymes are its targets? (2 points) What activates it [hormone (pathway activator) and enzyme (immediate activator)]? (2 points) Hormone: Enzyme:arrow_forward
- A.What is the Result of the sodium potassium ATPase activity in the cell? B.What mechanism does the sodium potassium ATPase use to achieve this result? C. Why is the above-mentioned change in affinity critical for the sodium potassium ATPase to perform its function?arrow_forward1. a. Calculate the physiological DG of the reaction shown below at 37°C, as it occurs in the cytosol ofneurons, with phosphocreatine at 4.7 mM, creatine at 1.0 mM, ADP at 0.73 mM, and ATP at 2.6mM. The standard free energy change for the overall reaction is –12.5 kJ/mol. Phosphocreatine + ADP ® creatine + ATP b. The enzyme phosphoglucomutase catalyzes the conversion of glucose 1-phosphate to glucose6-phosphate. Calculate the standard free energy change of this reaction if incubation of 20 mMglucose 1-phosphate (no glucose-6 phosphate initially present) yields a final equilibrium mixtureof 1.0 mM glucose 1-phosphate and 19 mM glucose 6-phosphate at 25°C and pH 7.0. c. If the rate of a nonenzymatic reaction is 1.2 x 10–2 μM s–1, what is the rate of the reaction at 37℃ inthe presence of an enzyme that reduces the activation energy by 30.5 kJ/mol?arrow_forwardIntravenous injection of KCl could be fatal. Why?arrow_forward
- Which statements are true of an inhibitor that binds the active site of an enzyme? Select all that apply, there may be one correct answer or several. a These inhibitors are a kind of allosteric regulator that decreases enzyme activity. b Adding more substrate can reduce the effect of these inhibitors. c These inhibitors compete with the substrate for the active site of the enzyme. d These inhibitors increase the rate of enzyme activity. e These inhibitors function by changing the shape of the enzyme, stopping if from binding to substrate.arrow_forwarda. Calculate how many carbons from an original glucose molecule will enter into the TCA cycle? (please provide your answer in number like 1, 2, 3) b. Based on the question you answered above what happens to the carbons that do not enter the cycle (write what it converts into)?arrow_forwarda. What is the efficiency of the metabolic conversion of palmitic acid to ATP? b. Compute the number of grams of palmitic acid that would have to be metabolized to provide the heat to warm a swimmer from whose skin 61 g of water evaporates.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education