
Concept explainers
(a)
Interpretation:
The structure and the stereochemistry of products formed by the reaction,
Concept introduction:
The addition of
Answer to Problem 7.55AP
The products formed by the reaction,
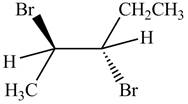
The stereo isomeric products that are formed are in the same amounts. Therefore, the racemic mixture is obtained.
Explanation of Solution
The reaction of
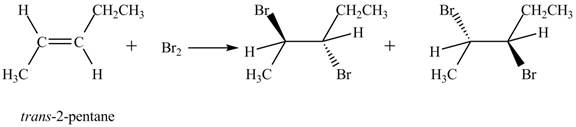
Figure 1
The products obtained are in equal ratio. Therefore, the racemic mixture is obtained.
The products formed by the reaction
(b)
Interpretation:
The structure and the stereochemistry of products formed by the reaction,
Concept introduction:
The addition of
Answer to Problem 7.55AP
The products formed by the reaction
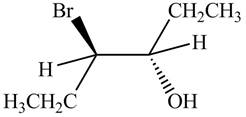
The stereo isomeric products that are formed are in the same amounts. Therefore, the racemic mixture is obtained.
Explanation of Solution
The reaction of
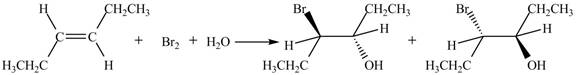
Figure 2
The products obtained are in equal ratio. Therefore, the racemic mixture is obtained.
The products formed by the reaction
(c)
Interpretation:
The structure and the stereochemistry of products formed by the reaction
Concept introduction:
The addition of
Answer to Problem 7.55AP
The products formed by the reaction
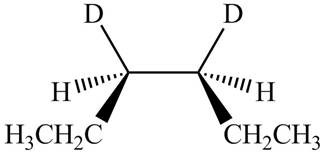
The stereoisomeric product that is obtained by the
Explanation of Solution
The reaction of
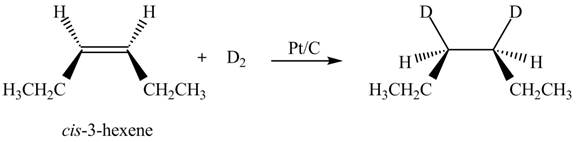
Figure 3
Therefore, the compound obtained is meso compound.
The products formed by the reaction
The stereo isomeric product that is obtained by the
(d)
Interpretation:
The structure and the stereochemistry of products formed by the reaction
Concept introduction:
The addition of
Answer to Problem 7.55AP
The product formed by the reaction,
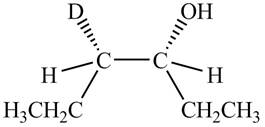
The stereo isomeric products that are formed are in the same amounts. Therefore, the racemic mixture is obtained.
Explanation of Solution
The reaction of
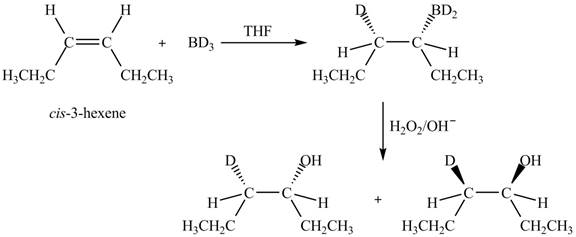
Figure 4
The products obtained are in equal ratio. Therefore, the racemic mixture is obtained.
The products formed by the reaction
The stereo isomeric products that are formed are in the same amounts.
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry
- Reaction of but-1-ene with HBr gives two products in unequal amounts. In each case, identify the two products, state which is the major product, explain why it is the major product and give the mechanism for its formation.arrow_forward7A Write the possible products of the following reactions, the mechanism by which they were formed. mentioning Br CH3OH CH3ONaarrow_forwardCompound X has the molecular formula C7H14. Hydrogenation of compound X produces 2,4-dimethylpentane. Hydroboration-oxidation of compound X produces a racemic mixture of 2,4-dimethyl-1-pentanol. Predict the major product(s) obtained when compound X is treated with aqueous acid (H3O*). Draw all the substrates, reagents, and productsarrow_forward
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardWhen 2-pentene is treated with Cl2 in methanol, three products are formed. Account for the formation of each product (you need not explain their relative percentages).arrow_forwardBicyclo-2,5-heptadiene can be prepared in two steps from cyclopentadiene and vinyl chloride. Provide a mechanism for each step.arrow_forward
- 5. Compound A, C 10H 18O, undergoes reaction with dilute H 2SO 4 at 50 °C to yield a mixture of two alkenes, C 10H 16. The major alkene B, gives only cyclopentanone after ozone treatment followed by reduction with zinc in acetic acid. Which of the following reactions are correct.arrow_forwardThe optically active (2R)-2-phenyl-2-butanol reacts in hydrochloric acid to form haloalkanes and alkenes. The substitution reaction is reported to occur with 100% racemization. Give the structures of the enantiomers that form during the substitution and indicate how much of each is formed. Propose a mechanism for the substitution reaction to yield the R product. What reagents would you use to achieve 100% retention of configuration. Two geometric isomers are obtained during the elimination reaction. Explain mechanistically which alkene will be the main product.arrow_forwardQuestão 10A certain hydrocarbon had the molecular formula C16H26 and contained two triple bonds. Ozonolysis resulted in CH3(CH2)4CO2H and HO2CCH2CH2CO2H as the only products. What is the reasonable structure for this hydrocarbon? Hexadec-6,10-dino undec-1,5-dino Hept-1,5-dino hex-1,5-dino naharrow_forward
- Which of the following alkenes is most likely to undergo rearrangement upon acid-catalyzed hydration (treatment with aqueous H,SO4)? HC=CH2 2 3 0 1 O 2 O 3 0 4arrow_forwardPropose a synthesis for the systemic agricultural fungicide tridemorph from dodecanoic acid (lauric acid), propene, and a one-carbon building block. How many stereoisomers are possible for tridemorph? CH3 →CH3(CH2)10COOH + CH3CH=CH, H3C (CH2) 12CH3 Tridemorph Dodecanoic acid Propene (Lauric acid)arrow_forwardCompound A, C 10H 18O, undergoes reaction with dilute H 2SO 4 at 50 °C to yield a mixture of two alkenes, C 10H 16. The major alkene B, gives only cyclopentanone after ozone treatment followed by reduction with zinc in acetic acid. Which of the following reactions are correct. Can be more than one answerarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

