
Practice calculating dilutions using the following problems.
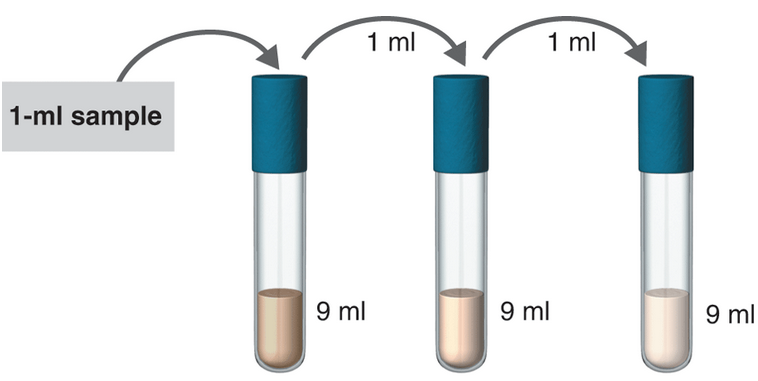
- Dilution:
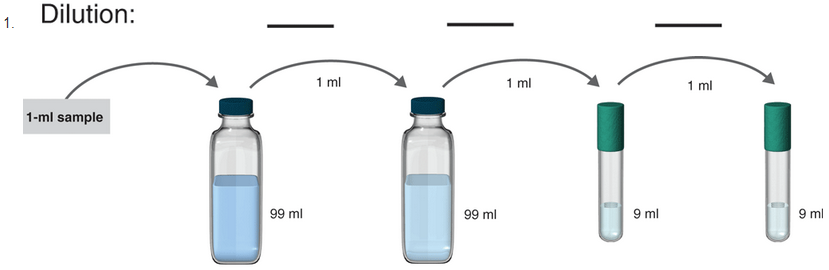
To determine:
The serial dilution in the given samples.
Introduction:
The stepwise dilution of a solution is termed as a serial dilution. Generally, the dilution factor at each stage of serial dilution is constant and leads to geometric progression in a logarithmic manner of the concentration of the sample. Serial dilutions are used to dilute the given substance at a very high level and serial dilutions are also used in the formation of concentration curves.
Explanation of Solution
The samples where serial dilution is occurring is shown below:
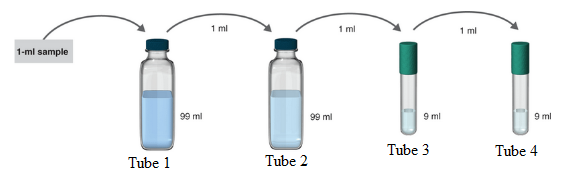
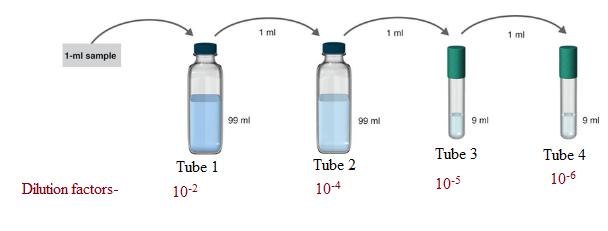
1 ml of the sample is mixed in 99ml solvent. So, the total solution is of 100 ml.
Hence, in tube 1 the serial dilution is of 1:100.
Now, 1 ml of the sample from tube 1 is poured to 99 ml solvent in tube 2. So, the total volume of the solution in tube 2 becomes 100 ml.
Hence, in tube 2 the serial dilution is now =
Now, 1 ml of the sample from tube 2 is poured to 9 ml solvent in tube 3. So, the total volume of the solution in tube 3 becomes 10 ml.
Hence, in tube 3 the serial dilution is now =
Now, 1 ml of the sample from tube 3 is poured to 9 ml solvent in tube 4. So, the total volume of the solution in tube 4 becomes 10 ml.
Hence, in tube 4 the serial dilution is now =
The samples where serial dilution is occurring is shown below:
Want to see more full solutions like this?
Chapter B Solutions
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
- Calculate the CFU present in 150 mL water sample and b) dilution factor up to 5. Consider the following dilution scheme.arrow_forwardCalculate the elution volume of blue dextran (blue), myoglobin (red), and Bromocresol purple (purple) in the gel filtration chromatography below with a flow rate of 2.5 mL/min. (Take note that the black colored test tubes represent colorless eluents)arrow_forwardShow workflow process of producing a Liquid Dosage Form through Extemporaneous CompoundingUse diagrams (boxes and arrows) to signify stepwise and significant procedures.arrow_forward
- What is your overall dilution factor if you complete 3 serial dilutions using a 100-fold dilution each time? (Show your work)arrow_forwardCalculate the volume of BSA stock that will be required to make the standard solutions needed to create the BSA standard curve. Be sure to show your work and include the volume of 0.02 M phosphate buffer required to reach a final volume of 1 mL. From a 2,000 μg/mL BSA stock, create 1 mL of each of the following stock solutions in 0.02 M phosphate buffer using individual microcentrifuge tubes: 50 μg/mL, 250 μg/mL, 500 μg/mL, 1,000 μg/mL, 1,250 μg/mL, 1,500 μg/mL. Be sure to properly label all the microcentrifuge tubes before creating the standards.arrow_forwardSketch a potentiometric biosensor measurementarrow_forward
- Consider the titration of 50.00 mL of 0.160 M NH3 with 0.200 M HCl. Calculate the pH at the following volumes of the titrant: VT = 0.00 (Initial Stage) VT = 20.00 VT = 30.00 VT = 40.00 (Equivalence Point Stage) VT = 45.00 Kb = 1.76 x 10-5arrow_forwardTwo students independently determine the volume of water delivered by a 10.00-mL pipet. Each student takes 8 measurements, then computes the average volume delivered and the standard deviation. The results are tabulated below. Average St. Dev. Student A 10.4 mL ±0.7 mL Student B 10.00 mL ±0.02 mL Which statement best describes the results? 1)A: good precision, poor accuracy. B: poor precision, good accuracy. 2)A: poor precision, good accuracy. B: good precision, poor accuracy. 3)A: poor precision, poor accuracy. B: good precision, good accuracy. 4)A: good precision, good accuracy. B: good precision, good accuracy. 5)A: poor precision, poor accuracy. B: poor precision, poor accuracy.arrow_forwardE. How would you make 8 mL of Wash Buffer (1X Lysis Buffer plus 30 mM Imidazole) from stocks of 2X Lysis buffer and 4 Imidazole?arrow_forward
- Exercise 6.1 Concentration & Dilution Data and Questions Name 1. Dilution Table Desired Desired Initial Volume of Stock or Volume of Concentration Volume Concentration Previous Dilution Distilled Water (V2) (C1) (V1), uL (V2- V1), µL (C2) 100 mg/dL 1.2 mL 200 mg/dL 75 mg/dL 1.2 mL 50 mg/dL 1.2 mL 25 mg/dL 1.2 mLarrow_forwardOther Math 1300mL N/Saline 0.9% is running at a 6 hourly rate, which commenced at 1500hrs. At 1630hrs the doctor requests for the remaining volume to be run over 3 hours. Calculate the mL per hour for the remaining volume.arrow_forwardCompute for the CFU/ml of drinking water. Is this Passed or Failed based on the Philippine National Standards for Drinking Water for Heterotrophic Plate Count (HPC)?arrow_forward

