
Concept explainers
(a)
Interpretation: The structure for given NMR spectra should be predicted.
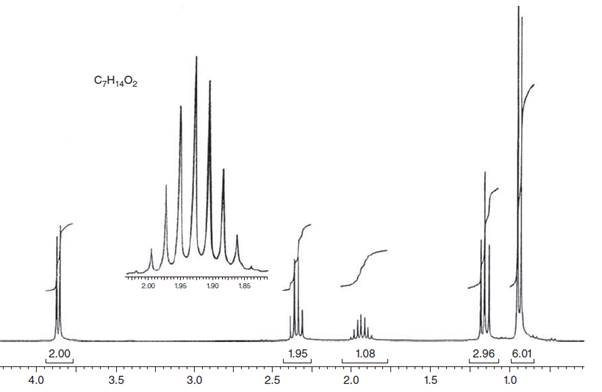
Concept Introduction:
NMR stands for nuclear magnetic resonance. A given compound contains different types of H-signals due to the presence of different magnetic environment within a molecule.
The number of signals obtained by the proton can be determined by spitting rule (n + 1) where n represents the number of the adjacent protons.
(b)
Interpretation: The structure for given NMR spectra should be predicted.
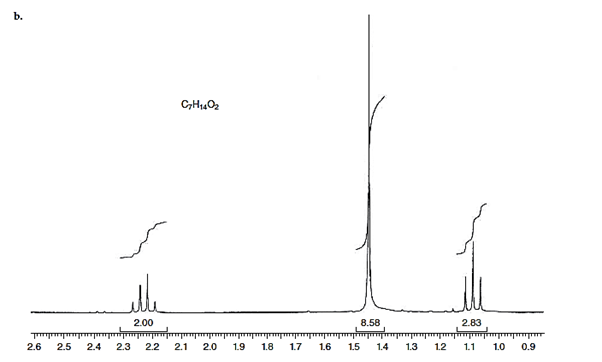
Concept Introduction:
NMR stands for nuclear magnetic resonance. A given compound contains different types of H-signals due to the presence of different magnetic environment within a molecule.
The number of signals obtained by the proton can be determined by spitting rule (n + 1) where n represents the number of the adjacent protons.
(c)
Interpretation: The structure for given NMR spectra should be predicted.
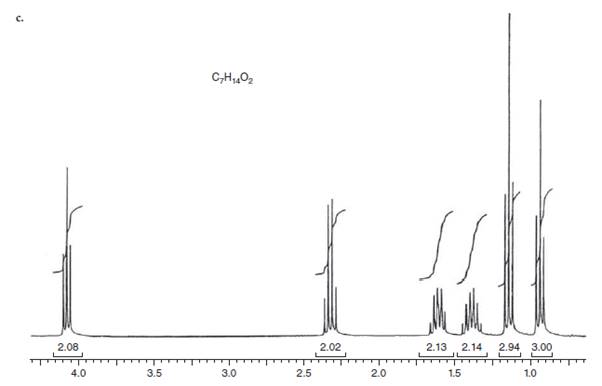
Concept Introduction:
NMR stands for nuclear magnetic resonance. A given compound contains different types of H-signals due to the presence of different magnetic environment within a molecule.
The number of signals obtained by the proton can be determined by spitting rule (n + 1) where n represents the number of the adjacent protons.
Trending nowThis is a popular solution!

Chapter 95 Solutions
EBK A SMALL SCALE APPROACH TO ORGANIC L
- What is the structure of an unknown compound with molecular formula C6H15N that gives the following 1H NMR absorptions: 0.9 (singlet, 1 H), 1.10 (triplet, 3 H), 1.15 (singlet, 9 H), and 2.6 (quartet, 2 H) ppm?arrow_forwardA compound with the molecular formula C4H₁1N has the following ¹H NMR spectrum, in which the relative integration values are illustrated with step curves. Which of the following is the correct number of protons giving rise to each signal? al O2H, 3H, 6H Ο 1Η, 4Η, 6Η O 2H, 4H, 5H O 3H, 4H, 5H PPMarrow_forwardThe 'H NMR spectrum of compound A (C3H100) has four signals: a multiplet at 8 = 7.25-7.32 ppm (5 H), a singlet at d = 5.17 ppm (1 H), a quartet at d = 4.98 ppm (1 H), and a doublet at ô = 1.49 ppm (3 H). There are 6 signals in its 13C NMR spectrum. The IR spectrum has a broad absorption in the -3200 cm-1 region. Compound A reacts with KMNO4 in a basic solution followed by acidification to give compound B with the molecular formula C7H6O2. Draw structures for compounds A and B.arrow_forward
- The 1H NMR spectra of two carboxylic acids with molecular formula C3H5O2Cl are shown below. Identify the carboxylic acids. (The “offset” notation means that the farthest-left signal has been moved to the rightby the indicated amount to fit on the spectrum; thus, the signal at 9.8 ppm offset by 2.4 ppm has an actual chemical shift of 9.8 + 2.4 = 12.2 ppm.)arrow_forwardidentify the compound with molecular formula C2H6O that gives this 1H NMR spectrum.arrow_forwardWhich of the protons in the following molecule appear at the highest 8-value in the ¹H NMR spectrum? Explain your reasoning. H₂ iii iv H CH3 i y HCarrow_forward
- Following are the 'H and 13C NMR spectra for each of three isomeric ketones with formula C7H14O. Determine a structure to each pair of spectra and assign each H and C. Carbon spectrum А C,H140 Carbon spectrum В C;H140 CDCI3 200 150 100 50 Proton spectrum CDC13 A C,H140 200 150 100 50 1.96 2.00 2.91 3.0 2.5 2.0 1.5 1.0 0.5 0.0 211.04 -44.79 –17.39 – 13.78 -218.40 - 38.85 –18.55arrow_forwardThe 1H NMR spectra of two carboxylic acids with molecular formula C3H5O2Cl are shown below. Identify the carboxylic acids. (The “offset” notation means that the farthest-left signal has been moved to the right by the indicated amount to fit on the spectrum; thus, the signal at 9.8 ppm offset by 2.4 ppm has an actual chemical shift of 9.8 + 2.4 = 12.2 ppm.)arrow_forwardThe only organic compound obtained when compound Z undergoes the following sequence of reactions gives the 1H NMR spectrum shown. Identify compound Z.arrow_forward
- The H1H1 NMR spectrum shown corresponds to an unknown compound with the molecular formula C6H10C6H10. There are no strong IR bands between 2100 and 2300 or 3250 and 3350 cm−1. Deduce and draw the structure of the molecule that corresponds to the spectrum.arrow_forwardFollowing is a 1H-NMR spectrum of 2-butanol. Explain why the CH2 protons appear as a complex multiplet rather than as a simple quintet.arrow_forwardpunqe You will characterize your product by 'H NMR spectroscopy. How many different proton environments do you expect your product compound to exhibit? How many "C environments would you expect to see in the "C NMR spectrum? OS IS TS ES YS SS 9S LS S 6S 09 19 19 99 59 99 L9 19 69 OL IL TL EL YL SL YL LL L 6L O1 I'8 X: parts per Million: Protonarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

