
What are the common and systematic names of the following ethers?
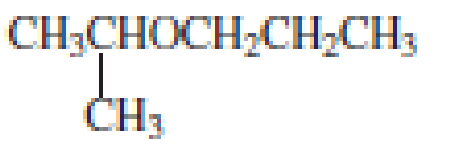
- a. CH3CH2CH2CH2OCH2CH3
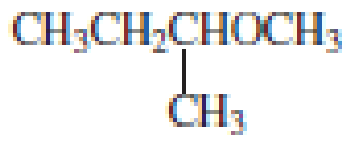
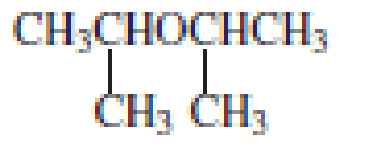
(a)
Interpretation:
The systematic name and the common name of the given ether have to be given.
Concept introduction:
IUPAC naming of Ether compounds:
An ether group consists of oxygen atom attached between two carbon chains. The shorter of two chains becomes the first part of the name with ‘ane’ changes to ‘oxy’ and the longer alkane becomes the suffix of the name of the ether.
When the Oxygen is not at the terminal position of main chain of alkane, then the shorter alkyl group and the ether group together are treated as a side chain and prefixed with its position of bonding on the main chain.
Common name for ether compounds:
- The name of the groups attached to the oxygen has to be given in alphabetical order followed by the word ether.
Explanation of Solution
The structure of the compound is given below:
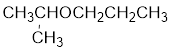
From the structure of the compound, it is understood that the compound is an ether with three carbons in the parent chain. Hence, the parent chain will be propane. The numbering of the longest chain will be in such a way to get the carbon atom attached to the oxygen the possible lowest number.
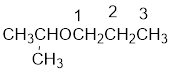
The shorter alkyl group attached to the oxygen is the isopropyl group.
Therefore, systematic name of the compound is given below:
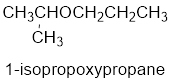
The common name of the compound can be identified by naming the alkyl groups attached to the oxygen followed by the word ether. In the given compound, one iso propyl group and one propyl group is attached to oxygen.
Therefore, common name of the compound is given below:
Propyl isopropyl ether
(b)
Interpretation:
The systematic name and the common name of the given ether has to be given.
Concept introduction:
IUPAC naming of Ether compounds:
An ether group consists of oxygen atom attached between two carbon chains. The shorter of two chains becomes the first part of the name with ‘ane’ changes to ‘oxy’ and the longer alkane becomes the suffix of the name of the ether.
When the Oxygen is not at the terminal position of main chain of alkane, then the shorter alkyl group and the ether group together are treated as a side chain and prefixed with its position of bonding on the main chain.
Common name for ether compounds:
- The name of the groups attached to the oxygen has to be given in alphabetical order followed by the word ether.
Explanation of Solution
The structure of the compound is given below:
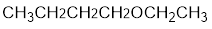
From the structure of the compound, it is understood that the compound is an ether with four carbons in the parent chain. Thus the parent chain will be butane. The numbering of the longest chain will be in such a way to get the carbon atom attached to the oxygen the possible lowest number.
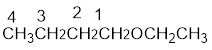
The shorter alkyl group attached to the oxygen that is in the first carbon is the ethyl group and therefore, the prefix will be “ethoxy”.
Therefore, the systematic name of the compound is given below:
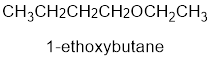
The common name of the compound can be identified by naming the alkyl groups attached to the oxygen followed by the word ether. In the given compound, one ethyl group and one butyl group is attached to oxygen.
Therefore, common name of the compound is given below:
butyl ethyl ether.
(c)
Interpretation:
The systematic name and the common name of the given ether has to be given.
Concept introduction:
IUPAC naming of Ether compounds:
An ether group consists of oxygen atom attached between two carbon chains. The shorter of two chains becomes the first part of the name with ‘ane’ changes to ‘oxy’ and the longer alkane becomes the suffix of the name of the ether.
When the Oxygen is not at the terminal position of main chain of alkane, then the shorter alkyl group and the ether group together are treated as a side chain and prefixed with its position of bonding on the main chain.
Common name for ether compounds:
- The name of the groups attached to the oxygen has to be given in alphabetical order followed by the word ether.
Explanation of Solution
The structure of the compound is given below:
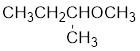
From the structure of the compound, it is understood that the compound is an ether with four carbons in the parent chain. Thus the parent chain will be butane. The numbering of the longest chain will be in such a way to get the carbon atom attached to the oxygen the possible lowest number.
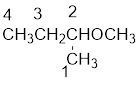
The oxygen is attached to the second carbon.
The shorter alkyl group attached to the oxygen is the methyl group and therefore, the prefix will be “methoxy”.
Therefore, the systematic name of the compound is given below:
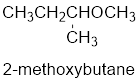
The common name of the compound can be identified by naming the alkyl groups attached to the oxygen followed by the word ether. In the given compound, one sec-butyl group and one methyl group is attached to oxygen.
Therefore, common name of the compound is given below:
Sec-butyl methyl ether.
(d)
Interpretation:
The systematic name and the common name of the given ether has to be given.
Concept introduction:
IUPAC naming of Ether compounds:
An ether group consists of oxygen atom attached between two carbon chains. The shorter of two chains becomes the first part of the name with ‘ane’ changes to ‘oxy’ and the longer alkane becomes the suffix of the name of the ether.
When the Oxygen is not at the terminal position of main chain of alkane, then the shorter alkyl group and the ether group together are treated as a side chain and prefixed with its position of bonding on the main chain.
Common name for ether compounds:
- The name of the groups attached to the oxygen has to be given in alphabetical order followed by the word ether.
Explanation of Solution
The structure of the compound is given below:
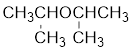
From the structure of the compound, it is understood that the compound is an ether with three carbons in the parent chain. Hence, the parent chain will be propane.
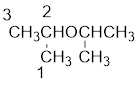
The shorter alkyl group attached to the oxygen that is in the second carbon is the isopropyl group and therefore, the prefix of the name will be “isopropoxy”.
Therefore, systematic name of the compound is given below:
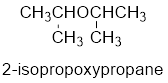
The common name of the compound can be identified by naming the alkyl groups attached to the oxygen followed by the word ether. In the given compound, diisopropyl groups are attached to oxygen.
Therefore, common name of the compound is given below:
disopropyl ether.
Want to see more full solutions like this?
Chapter 9 Solutions
Essential Organic Chemistry, Global Edition
- 1. Name the following ether. 2. Name the following thiol. C C-C-C-C-0-C-C -9-9-⁹ C-C-C-C-C-S-H C-C-C-C 3. Will the ether H bond with water? Will the thiol H bond with water?arrow_forward2. Draw the structures of the starting organic compounds that can be used to make the following ethers. ontarrow_forwardDefine Preparation of Ethers ?arrow_forward
- Give the IUPAC name for each compound. OH a. CH3CH(CH₂)4CH3 (select) OH (CH3CH₂)2CHCHCH₂CH3 (select) b. C. d. CH3 OH (select) (select) (select) OH (select) (select) (select)arrow_forwardWhat is the IUPAC name of the following compound? Br CH3 H3C O A. 5-Bromo-2-methylphenyl ethanoate B. 3-Bromo-6-methylphenyl ethanoate C. 4-Bromo-2-{oxy-(1-oxoethyl)} toluene EO D. Methyl-5-bromo-2-methyl benzoatearrow_forwardWhat is the IUPAC name of the following compound? OH Br N=C O A. 3-bromo-4-hydroxybenzonitrile B. 2-bromo-4-cyanophenol C. 2-bromo-4-cyano-1-hydroxybenzene D. 2-bromo-4-cyanobenzanolarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


