
Concept explainers
Draw Lewis structures for the following molecules and ions:
Interpretation:
The Lewis structures of the given molecules and ions are to be drawn.
Concept introduction:
In Lewis dot symbol, for each element, dots are mentioned around the symbol of an atom.
In Lewis dot symbol, valence electrons are represented by dots.
Dots are placed above and below as well as to the left and right of symbol.
Number of dots is important in Lewis dot symbol but not the order in which the dots are placed around the symbol.
In writing symbol pairing is not done until absolutely necessary.
For metals, the number of dots represents the number of electrons that are lost when the atom forms a cation.
For second period nonmetals, the number of unpaired dots is the number of bonds the atom can form.
Atomic ions can also be represented by dot symbols, by simply adding (for anions) and subtracting (for cations) the appropriate number of dots from Lewis dot symbol.
Lewis structure is the representation of bonding and nonbonding electron pairs present in the outermost shell of all atoms present in the molecule.
The number of bonds formed by an atom in the molecule is determined by the valence electron pairs.
Answer to Problem 47QP
Solution:
a)
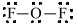
b)
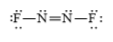
c)
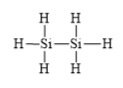
d)
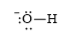
e)
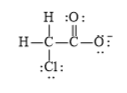
f)
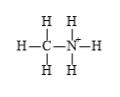
Explanation of Solution
a)
The electronic configuration of oxygen and fluorine in
The oxygen atom contains fourvalence electrons in its
The Lewis structure of
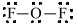
b)
The electronic configuration of nitrogen and fluorine in
The nitrogen atom contains three valence electrons in its
The Lewis structure of
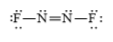
c)
The electronic configuration of silicon and hydrogen in
The silicon atom has a tendency to form four bonds because of the presence of four valence electrons in its outermost shell and hydrogen has a tendency to form one bond because of the presence of one electron in its outermost shell.
The Lewis structure is as follows:
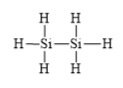
d)
The electronic configuration of oxygen and hydrogen in
The oxygen atom contains four valence electrons in its
The Lewis structure of
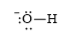
e)
The electronic configuration of oxygen, carbon, chlorine, and hydrogen in
The carbon atom has a tendency to form four bonds because of the presence of four electrons in its outermost shell, hydrogen has a tendency to form one bond because of the presence of one electron in its outermost shell, chlorine has atendency to form one bond because of the presence of five electrons in its
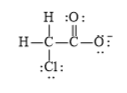
f)
The electronic configuration of carbon, nitrogen, and hydrogen in
The carbon atom has a tendency to form four bonds because of the presence of four electrons in its outermost shell, hydrogen has a tendency to form one bond because of the presence of one electron in its outermost shell, and nitrogen has a tendency to form four bonds due to the presence of three electrons in its

Want to see more full solutions like this?
Chapter 8 Solutions
Chemistry
- Write Lewis dot symbols for the following atoms and ions: (a) I (b) I-(c) S(d) S2-(e) P (f) P3-(g) Na (h) Na+(i) Mg (j) Mg2+arrow_forwardWrite Lewis structures for the following:(a) O2(b) H2CO(c) AsF3(d) ClNO(e) SiCl4(f) H3O+(g) NH4+(h) BF4−(i) HCCH(j) ClCN(k) C22+arrow_forwardDraw the Lewis structures and determine which of these molecules is least likely to exist. (A) H3S+ (B) H3O+ (С) НзО- (D) H3S- (E) OH- Draw all 5 structures with 1 central atom. All of the other atoms are attached to the central atom.arrow_forward
- In which of the following compounds does hydrogen bear a partial negative charge: (a) CH4, (b) NH3, (c) H2O, (d) SiH4 or (e) H2S?arrow_forwardWrite Lewis structures for the following molecules or ions:(a) SbH3(b) XeF2(c) Se8 (a cyclic molecule with a ring of eight Se atoms)arrow_forwardWrite Lewis structures for the following: (c) C2F6 (contains a C¬C bond), (d) AsO3 3 -, (e) H2SO3 (H is bonded to O), (f) NH2Cl.. Arrange the bonds in each of the following sets in order of increasing polarity: (a) C¬F, O¬F, Be¬F; (b) O¬Cl, S¬Br, C¬P; (c) C¬S, B¬F, N¬O. What is the Lewis symbol for each of the following atoms or ions? (a) K, (b) As, (c) Sn2 + , (d) N3 Write electron configurations for the following ions and determine which have noble-gas configurations: (a) Cd2+, (b) P3-, (c) Zr4+arrow_forward
- Draw the Lewis structures for each of the following ions or molecules. Give the number of electrons in each species. Remember to enclose ion s in square brackets with the charge as a superscript outside the right bracket. (a) SO 2 (b) XeO 2 F 2 (Xe is the central atom) (c) ClF 3 (d) ClO 2 F (Cl is the central atom) (e) BrO 4 -arrow_forwardDraw Lewis structures for the following: (a) SiH4, (b) CO,(c) SF2, (d) H2SO4 (H is bonded to O), (e) ClO2-, (f) NH2OH.arrow_forwardWrite Lewis structures for the following molecules or ions. (Assign lone pairs, radical electrons, and atomic charges where appropriate.) (a) SbO43− (b) ICl6− (c) SO32- (d) HOBrOarrow_forward
- (a) Describe the molecule xenon trioxide, XeO3, using four possible Lewis structures, one each with zero, one, two, or three Xe—O double bonds. (b) Do any of these resonance structures satisfy the octet rule for every atom in the molecule? (c) Do any of the four Lewis structures have multiple resonance structures? If so, how many resonance structures do you find? (d) Which of the Lewis structures in (a) yields the most favorable formal charges for the molecule?arrow_forwardConsider the following compounds: CaCl 2 , CaI 2 , and MgCl 2 . Answer the following questions based on expected periodic trends: (a) Which is expected to have t he shortest ionic bonds? (b) Which is expected to have the highest lattice energy? (c) Which is expected to have the lowest melting point?arrow_forwardWrite the Lewis symbol for atoms of each of the following elements:(a) Al, (b) Br, (c) Ar, (d) Sr.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

