
(a)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.
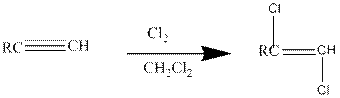
If excess halides are present, a second addition reaction also occurs.
(b)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
(c)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
(d)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
(e)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
(f)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Organic Chemistry (8th Edition)
- Draw structure for the major reaction products of each of the following reactionsarrow_forwardWhat is the major product of the following reactionarrow_forwardCalculate the AH of a reaction with bond strengths of bonds broken equal to 60 kcal/mol and 107 kcal/mol and bond strengths of bond formed equal to 110 kcal/mol and 88 kcal/mol. -245 kcal/mol -31 kcal/mol 151 kcal/mol O 31 kcal/molarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





