
Concept explainers
Draw Lewis structures and give the other information requested for the following molecules: (a) BF3, Shape: planar or nonplanar? (b) ClO3− Shape: planar or nonplanar? (c) HCN. Polar or nonpolar? (d) OF2. Polar or nonpolar? (c) NO2. Estimate the ONO bond angle.
a)
Interpretation: The Lewis structures of the given molecules should be drawn. To identify whether
Concept Introduction:
- Molecular geometry is the spatial arrangement of atoms in a molecule. It is the three dimensional arrangement of bonded atoms. Many chemical, physical and even biological properties depend on molecular geometry.
- Bond angle measured that made between two nearby bonds. The angles between two adjacent bonds are known as bond angle.
- Using VSEPR theory and Lewis structure, the exact geometry of a molecule can be obtained.
- In VSEPR, the geometry of the molecule is explained based on minimizing electrostatic repulsion between the molecules’ valence electrons around a central atom
- Lewis structures is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electronsthat may exist in the molecule.
- While formation of a bond, there is a chance that electronegativity between the atoms are high. It tends to make the bond partially ionic and are called polar molecule. When the electronegativity between two atoms is similar, sharing of electron in the bond is equal and is called nonpolar molecules.
Answer to Problem 7.105QP
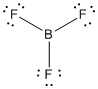 is the Lewis structure of
is the Lewis structure of
Explanation of Solution
Lewis structure of

Here boron is taken as the central atom with three terminal fluorine atoms since electronegativity of boron is less than fluorine. The total number of valence electrons is found to be 24, where each of the fluorine atoms contributes 6 electrons whereas boron contributes 3 electrons.
The 18 electrons getting after reducing two electrons for each bond from the total valence electron are distributed on terminal fluorine atom to complete the octet. After the distribution of electrons on the terminal atoms, each fluorine atoms gets 3 pairs of electrons.
Here the central atom boron atom does not have any lone pair of electrons whereas the terminal fluorine atoms have 3 pairs of electron. It is a
b)
Interpretation: The Lewis structures of the given molecules should be drawn. To identify whether
Concept Introduction:
- Molecular geometry is the spatial arrangement of atoms in a molecule. It is the three dimensional arrangement of bonded atoms. Many chemical, physical and even biological properties depend on molecular geometry.
- Bond angle measured that made between two nearby bonds. The angles between two adjacent bonds are known as bond angle.
- Using VSEPR theory and Lewis structure, the exact geometry of a molecule can be obtained.
- In VSEPR, the geometry of the molecule is explained based on minimizing electrostatic repulsion between the molecules’ valence electrons around a central atom
- Lewis structures is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electronsthat may exist in the molecule.
- While formation of a bond, there is a chance that electronegativity between the atoms are high. It tends to make the bond partially ionic and are called polar molecule. When the electronegativity between two atoms is similar, sharing of electron in the bond is equal and is called nonpolar molecules.
Answer to Problem 7.105QP
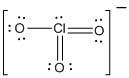 is the Lewis structure of
is the Lewis structure of
Explanation of Solution
Lewis structure of

Here chlorine is taken as the central atom with three terminal oxygen atoms. The total number of valence electrons is found to be 25 where each of the oxygen atoms contributes 6 electrons whereas chlorine contributes 5 electrons. The charge of the whole molecule is -1 making the total number of valence electrons 26.
The 20 electrons getting after reducing two electrons for each bond from the total valence electron are distributed on terminal oxygen atom to complete the octet. Remaining 2 electrons were distributed on central phosphorous atom. Since the atoms do not obey the octet rule two double bonds are made between chlorine and oxygen atoms.
In the case of
c)
Interpretation: The Lewis structures of the given molecules should be drawn. To identify whether
Concept Introduction:
- Molecular geometry is the spatial arrangement of atoms in a molecule. It is the three dimensional arrangement of bonded atoms. Many chemical, physical and even biological properties depend on molecular geometry.
- Bond angle measured that made between two nearby bonds. The angles between two adjacent bonds are known as bond angle.
- Using VSEPR theory and Lewis structure, the exact geometry of a molecule can be obtained.
- In VSEPR, the geometry of the molecule is explained based on minimizing electrostatic repulsion between the molecules’ valence electrons around a central atom
- Lewis structures is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electronsthat may exist in the molecule.
- While formation of a bond, there is a chance that electronegativity between the atoms are high. It tends to make the bond partially ionic and are called polar molecule. When the electronegativity between two atoms is similar, sharing of electron in the bond is equal and is called nonpolar molecules.
Answer to Problem 7.105QP
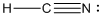 is the Lewis structure of
is the Lewis structure of
Explanation of Solution
Lewis structure of
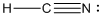
Here carbon is taken as the central atom with onenitrogen and one hydrogen atom in the terminal position since electronegativity of carbon is less. The total number of valence electrons is found to be 10, where each of the nitrogen atoms contributes 5 electrons whereas carbon contributes 4 electrons whereas hydrogen atom contributes only one electron.
The 6 electrons getting after reducing two electrons for each bond from the total valence electron are distributed on terminal nitrogen atom to complete the octet. Since the atoms do not obey the octet rule a triple bond is made between carbon and nitrogen atom.
In
d)
Interpretation: The Lewis structures of the given molecules should be drawn. To identify whether
Concept Introduction:
- Molecular geometry is the spatial arrangement of atoms in a molecule. It is the three dimensional arrangement of bonded atoms. Many chemical, physical and even biological properties depend on molecular geometry.
- Bond angle measured that made between two nearby bonds. The angles between two adjacent bonds are known as bond angle.
- Using VSEPR theory and Lewis structure, the exact geometry of a molecule can be obtained.
- In VSEPR, the geometry of the molecule is explained based on minimizing electrostatic repulsion between the molecules’ valence electrons around a central atom
- Lewis structures is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electronsthat may exist in the molecule.
- While formation of a bond, there is a chance that electronegativity between the atoms are high. It tends to make the bond partially ionic and are called polar molecule. When the electronegativity between two atoms is similar, sharing of electron in the bond is equal and is called nonpolar molecules.
Answer to Problem 7.105QP
Answer
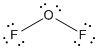 is the Lewis structure of
is the Lewis structure of
Explanation of Solution
Lewis structure of

Here oxygenis taken as the central atom with two terminal fluorine atoms since electronegativity of oxygen is less than fluorine. The total number of valence electrons is found to be 20, where each of the fluorine atoms contributes 7 electrons whereas oxygen contributes 6 electrons.
The 16 electrons getting after reducing two electrons for each bond from the total valence electron are distributed on silicon atom to complete the octet. After the distribution of electrons on the terminal atoms, each fluorine atoms gets 3 pairs of electrons. The remaining 4 electrons are distributed to the oxygen atom.
The central atom has two lone pairs thus it has four electron domains so it have bent geometry. Due to the difference in the electronegativity the bonds are polar. But due to the presence of lone pair on the central atom, the molecule is not symmetrical. The dipole vectors do not cancel each other thus having a total dipole moment on the molecule. So it is a polar molecule.
e)
Interpretation: The Lewis structures of the given molecules should be drawn. To identify whether
Concept Introduction:
- Molecular geometry is the spatial arrangement of atoms in a molecule. It is the three dimensional arrangement of bonded atoms. Many chemical, physical and even biological properties depend on molecular geometry.
- Bond angle measured that made between two nearby bonds. The angles between two adjacent bonds are known as bond angle.
- Using VSEPR theory and Lewis structure, the exact geometry of a molecule can be obtained.
- In VSEPR, the geometry of the molecule is explained based on minimizing electrostatic repulsion between the molecules’ valence electrons around a central atom
- Lewis structures is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electronsthat may exist in the molecule.
- While formation of a bond, there is a chance that electronegativity between the atoms are high. It tends to make the bond partially ionic and are called polar molecule. When the electronegativity between two atoms is similar, sharing of electron in the bond is equal and is called nonpolar molecules.
Answer to Problem 7.105QP
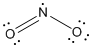 is the Lewis structure of
is the Lewis structure of
Explanation of Solution
Lewis structure of

Here nitrogen is taken as the central atom with two terminal oxygen atoms since electronegativity of oxygen is less than fluorine. The total number of valence electrons is found to be 20, where each of the fluorine atoms contributes 7 electrons whereas oxygen contributes 6 electrons.
The 12 electrons getting after reducing two electrons for each bond from the total valence electron are distributed on terminal oxygen atom to complete the octet. The remaining one electron is distributed to the nitrogen atom. Since the atoms do not obey the octet rule a double bond is made between one of the oxygen and nitrogen atom.
Want to see more full solutions like this?
Chapter 7 Solutions
Chemistry: Atoms First
- Predict die molecular structure and bond angles for each molecule or ion in Exercises 88 and 94. a. POCl3, SO42, XeO4, PO43, ClO4 b. NF3, SO32, PO33, ClO3 c.ClO2, SCl2, PCl2 d. Considering your answers to parts a, b, and c. what conclusions can you draw concerning the structures of species containing the same number of atoms and the same number of valence electrons? (O3), sulfur dioxide, and sulfur trioxide.arrow_forwardConsider the following compounds: CO2, SO2, KrF2, SO3, NF3, IF3, CF4, SF4, XeF4, PF5, TF5, and SCl6. These 12 compounds are all examples of different molecular structures. Draw the Lewis structures for each and predict the molecular structures. Predict the bond angles and the polarity of each. (A polar molecule has a net dipole moment, while a nonpolar molecule does not.) See Exercises 25 and 26 for the molecular structures based on the trigonal bipyramid and the octahedral geometries.arrow_forwardConsider the following compounds: CO2, SO2, KrF2, SO3, NF3, IF3, CF4, SF4, XeF4, PF5, IF5, and SCl6. These 12 compounds arc all examples of different molecular structures. Draw the Lewis structures for each and predict the molecular structure. Predict the bond angles and the polarity of each. (A polar molecule has a net dipole moment, while a nonpolar molecule docs not.) See Exercises 115 and 116 for the molecular structures based on the trigonal bipyramid and the octahedral geometries.arrow_forward
- In each of the following molecules, a central atom is surrounded by a total of three atoms or unshared electron pairs: SnCl2, BCl3, SO2. In which of these molecules would you expect the bond angle to be less than 120? Explain your reasoning.arrow_forwardMethyl isocyanate, CH3NCO, was made infamous in 1984when an accidental leakage of this compound from a storagetank in Bhopal, India, resulted in the deaths of about3800 people and severe and lasting injury to many thousandsmore. (a) Draw a Lewis structure for methyl isocyanate.(b) Draw a ball-and-stick model of the structure,including estimates of all the bond angles in the compound.(c) Predict all the bond distances in the molecule.(d) Do you predict that the molecule will have a dipolemoment? Explain.arrow_forward1) Why is it important to understand the molecular geometry or shape of molecules and ions in chemistry? H2Se CCl4 CH2Cl2 HCN NH3Cl+ NH2- CH3COCH3 CH3COOH 2) Among the eight molecules and ions studied in this experiment, (a) which molecule(s) or ion(s) are polar, and which ones are non-polar? (b) What is the difference between polar and non-polar molecules or ions? (c) What two factors dictate the polarity of the molecule or ion?arrow_forward
- B. ClF3 (1) Draw the valid Lewis structure, identify the (2) molecular group geometry, (3) electron group geometry, (4) polarity of the bonds, (5) over-all polarity, (6) number of lone pairs, and (7) number of bonding pairs.arrow_forwardDraw the resonance forms that fit the octet rule for sulfur trioxide, SO 3 . (a) How many sigma bonds are there? How many pi bonds? (b) What is the sulfur-oxygen bond order? (c) Arrange the following species in order of decreasing S-O bond strength (strongest first, weakestlast). SO, SO 3^2– , SO 3 , SO 2arrow_forwardTwo important industrial chemicals, ethene, C2H4, and propene, C3H6, are produced by the steam (or thermal) cracking process: 2C3H8(g) ⟶ C2H4(g) + C3H6(g) + CH4(g) + H2(g) For each of the four carbon compounds, do the following: (a) Draw a Lewis structure. (b) Predict the geometry about the carbon atom. (c) Determine the hybridization of each type of carbon atom.arrow_forward
- Two important industrial chemicals, ethene, C2H4, and propene, C3H6, are produced by the steam (or thermal) cracking process:2C3 H8(g) ⟶ C2 H4(g) + C3 H6(g) + CH4(g) + H2(g)For each of the four carbon compounds, do the following:(a) Draw a Lewis structure.(b) Predict the geometry about the carbon atom.(c) Determine the hybridization of each type of carbon atom.arrow_forwardPlease answer this question, Dont use cursive handwriting, answer in clear step by step solutions, For the following molecules, give the: Lewis diagram, name of shape, bond angles (DON'T use bond angles from vsepr chart), and polarity a) F2O b) SF6 Thank you!arrow_forward). Which of the following molecules contain polar covalent bonds? water, H2O carbon monoxide, CO fluorine, F2 nitrogen, N2arrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning




