
Concept explainers
Interpretation:
The curved arrow notation is to be drawn for the proton transfer between ammonia
Concept introduction:
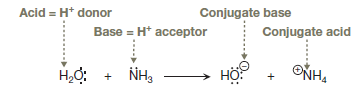
In a proton transfer reaction, a proton is transferred from a Bronsted–Lowry acid (proton donor) to a Bronsted–Lowry base (proton acceptor) in a single elementary step in which one bond is broken and another is formed simultaneously. The conjugate acid is the species that the base becomes after gaining a proton, and the conjugate base is the species that the acid becomes after losing a proton. The curved arrow notation shows the movement of valence electrons, not atoms. Each double-barbed curved arrow shows the movement of two valence electrons. To represent bond breaking, the tail of the arrow originates from the center of a bond whereas to represent bond formation, the head of the arrow points to an atom that forms the new bond, that is,
Answer to Problem 6.1P
The curved arrow notation for the proton transfer between ammonia
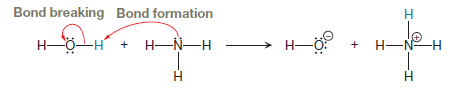
Explanation of Solution
The given proton transfer reaction is between ammonia
The bond breaking and bond formation involves only valence electrons, so first, we need to draw all valence electrons in the given two reactants. From this, it is clearly seen which electrons are involved in the reaction, both from the reactants and from the products as shown below:
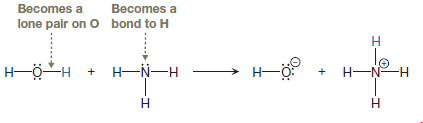
The appropriate movement of these valence electrons is shown by using curved arrow notations. One curved arrow is to be drawn from the lone pair on N to the H on water to illustrate the formation of

The curved arrow notation for the proton transfer of the given reaction is drawn on the basis of movement of valence electrons involved in bond breaking and bond formation.
Want to see more full solutions like this?
Chapter 6 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- When perchloric acid ionizes, it makes the perchlorate ion, ClO4. Draw the Lewis electron dot symbol for the perchlorate ion.arrow_forwardWrite equations that show H2PO4- acting both as an acid and as a base.arrow_forwardWhich of the terms weak, strong, monoprotic, diprotic, and triprotic characterize(s) each of the following acids? More than one term may apply in a given situation. a. H3PO4 b. H3PO3 c. HBr d. HC2H3O2arrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





