
What phase change is shown in the accompanying molecular art? Is energy absorbed or released during the process?
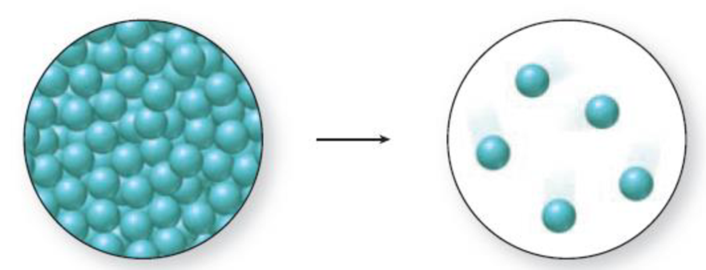
Interpretation:
The phase change accompanying the molecular art and the process of reaction (exothermic and endothermic) has to be given.
The given diagram is,
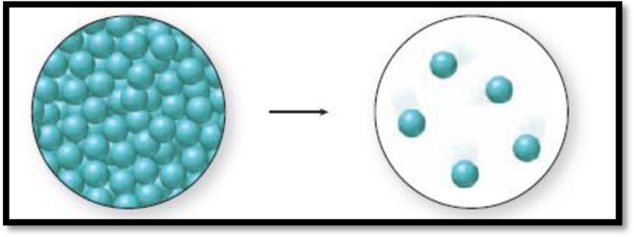
Figure 1
Concept introduction:
Endothermic Reactions:
The endothermic process is a term that describes a reaction where the system absorbs the energy from its surrounding in the form of heat. Few examples of endothermic process is photosynthesis, evaporating liquids, melting ice.
Exothermic reaction:
The exothermic reaction is the opposite of an endothermic reaction. It releases energy by light or heat to its surrounding. Few examples are neutralization, burning a substance, reactions of fuels, deposition of dry ice, respiration.
Vaporization:
Vaporization means conversion of a substance from the liquid phase to the gaseous phase.
Explanation of Solution
The given diagram is,
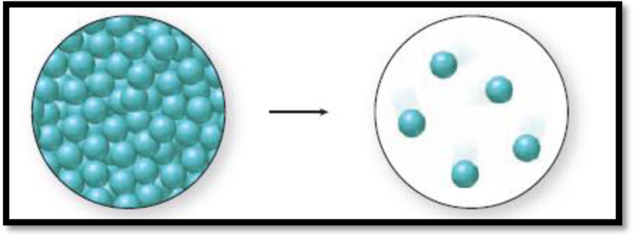
Figure 1
The change in phase can be identified by the distance between the spheres and the level of organization. A solid has closely packed spheres that are well organized, a liquid has closely packed but randomly arranged spheres, and a gas has randomly arranged spheres that are farther away from each other.
The diagram 1 represents a liquid and the diagram two represents a gas. Vaporization is the process of change from liquid to gas. Thus., the phase change accompanied in the figure 1 is a vaporization process and it is an endothermic process because liquid molecules absorb energy to go from liquid state to gaseous state.
Want to see more full solutions like this?
Chapter 4 Solutions
Principles of General, Organic, Biological Chemistry
- True or false? Mixtures always result in a chemical reaction because they consist of two or more substances and thus combine to create a new product.arrow_forwardA particulate-level illustration of the reaction AB+CDAD+CB is shown below. a Identify the reactants and products in this reaction. b Is the change shown chemical or physical? c Is the mass of the product particles less than, equal to, or greater than the mass of the reactant particles? d If the reaction takes place in a container that allows no energy to enter or leave, how does the total energy in the container after the reaction compare with the total energy in the container before the reaction?arrow_forwardaIs the process of boiling water exothermic or endothermic with respect to the water? bA charged object is moved closer to another object that has the same charge. The energy of the system changes. Is it a change in kinetic energy or potential energy? Is the energy changes an increase or a decrease?arrow_forward
- A book is held 6 feet above the floor and then dropped. Which statement is true? a.The potential energy of the book is converted to kinetic energy. b.The potential energy of the book is destroyed. c.Kinetic energy is created. d.The total energy of the system will not be conserved.arrow_forwardAs a child plays on a swing, at what point in her movement is her kinetic energy the greatest? At what point is potential energy at its maximum?arrow_forwardIf 100. J of heat energy is applied to a 25-g sample of mercury, by how many degrees will the temperature of the sample of mercury increase? (See Table 10.1.)arrow_forward
 World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





