
Concept explainers
(a)
Interpretation: Overall equation and the propagation steps of the mechanism for the chlorination of chloromethane to furnish dichloromethane should be written.
Concept introduction: The monochlorination performed with ultraviolet light proceeds via radical chain mechanism. Chlorine transforms
The fundamental radical chain mechanism is summarized in the illustration as follows:
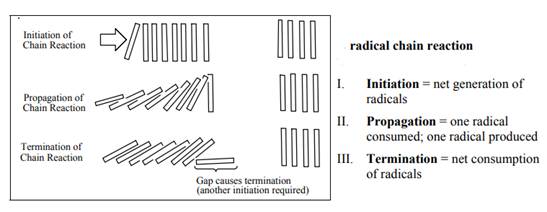
(b)
Interpretation: The formation of small amount of ethane in chlorination of methane should be determined.
Concept introduction: The monochlorination performed with ultraviolet light proceeds via radical chain mechanism. Chlorinetransforms
The fundamental radical chain mechanism is summarized in the illustration as follows:
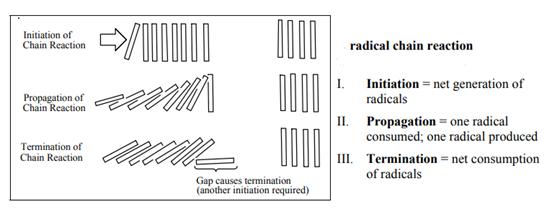
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Organic Chemistry: Structure and Function
- Define the Mechanism of the Radical Addition of HBr to an Alkene ?arrow_forwardPhotochemical chlorination of 2,2,4-trimethylpentane gives four isomeric monochlorides. (a) Write structural formulas for these four isomers. (b) The two primary chlorides make up 65% of the monochloride fraction. Assuming that all the primary hydrogens in 2,2,4-trimethylpentane are equally reactive, estimate the percentage of each of the two primary chlorides in the product mixture.arrow_forwardThe aerobic oxidation of para-xylene to terephthalic acid is an important process in industrial chemistry. Discuss why the oxidation of the second methyl group requires harsher conditions than the oxidation of the first methyl group. You should accurately reference all your bibliographic material.arrow_forward
- (b) Predict the suitable solvent (H2O or CH3COCH3) to increase the reaction of bromopropane (CH3CH2CH2B1) with sodium hydroxide (NaOH). Two reactions are shown below: NaOH, 55 °C CH;CH,CH,Br CH;CH,CH,OH + NaBr H,O (i) NaOH, 55 °C CH;CH,CH,Br CH;CH,CH,OH NaBr H,C CH (ii)arrow_forwardWhen exactly 1 mole of methane is mixed with exactly 1 mole of chlorine and light is shone on the mixture, a chlorination reaction occurs. The products are found to contain substantial amounts of di-, tri-, and tetrachloromethane, as well as unreacted methane. Explain how a mixture is formed from this stoichiometric mixture of reactants, and propose mechanisms for the formation of these compounds from chloromethane.arrow_forward(a) (i) 3-Methyl-2-butanol will react with sulphuric acid to give two isomeric alkenes in 3:1 proportions.(i) Write down the structures for these alkenes and assign appropriate systematic names to them. (ii) Name the most abundant isomer. (iii) Write down detailed mechanism for the formation of a minor product.arrow_forward
- A certain hydrocarbon had the molecular formula C18H30 and contained two triple bonds. Ozonolysis gave CH₂(CH₂)CO₂H and HO₂CCH₂CH₂CO₂H as the only products. Draw a reasonable structure for this hydrocarbon. Click and drag to start drawing a structure.arrow_forward(b) Compare combustion of Anthracene and Xylene in sufficient supply of air. Which one will produce more carbon rich sooty flame and why?arrow_forwardWrite the initiation, propagation, and termination steps for the monochlorination of cyclohexane.arrow_forward
- The rate of hydration of 2-butene and 2-methylpropene differ by several orders of magnitude. Please state which alkene is more rapidly hydrated and why.arrow_forwardA student reacts 2-pentene with H2(g) and chooses iron (Fe) as his catalyst. Identify the outcome the student will get from this reaction.arrow_forwardAlkyl halides can be reduced to alkanes by a radical reaction with tributyltin hydride, (C4H9)3SnH, in the presence of light (hv). Propose a radical chain mechanism by which the reaction might occur. The initiation step is the light-induced homolytic cleavage of the Sn-H bond to yield a tributyltin radical.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

