
To review:
The differences between the properties of water (H2O), ammonia (NH3), and methane (CH4), based on the hydrogen bonding in their structure. The heat of fusion for water, ammonia, and methane is given as 6.01, 5.66, and 0.94 kJ/mol, respectively. Also, determine increase or decrease in the density of ammonia in the form of ice (if generated), as compared to liquid ammonia.
Introduction:
The molecular mass of water, ammonia, and methane is almost equal, and all of them show tetrahedral geometry. The heat of fusion is the highest in water, decreases in ammonia, and is the least in methane. Water has a highly polar structure. The three molecules (water, ammonia, and methane) are sp3 hybridized and are tetrahedral instructure. However, there is a difference in the number of lone pairs in them, leading to an overall different geometry and different physical properties.
Explanation of Solution
Water has two hydrogen atom, shich are covalently bonded to one oxygen atom. The oxygen is sp3 hybridized. Due to the presence of twolone pairs, this molecule has a bent geometry. As a whole, a water molecule is polar and acts as a dipole. The electronegativity of oxygen is more than that of hydrogen, and thus, it bears a partial negative charge, whereas two hydrogen atoms beara partial positive charge. The hydrogen atoms in asingle water molecule are electrondeficient, and thus, tend to be attracted toward the oxygen atom of another water molecule. Thus, hydrogen bonds (intermolecular)act as bridges between neighboringwater molecules. One water molecule can form hydrogen bonds with four otherwater molecules.
The structures of thethreemolecules can be represented as
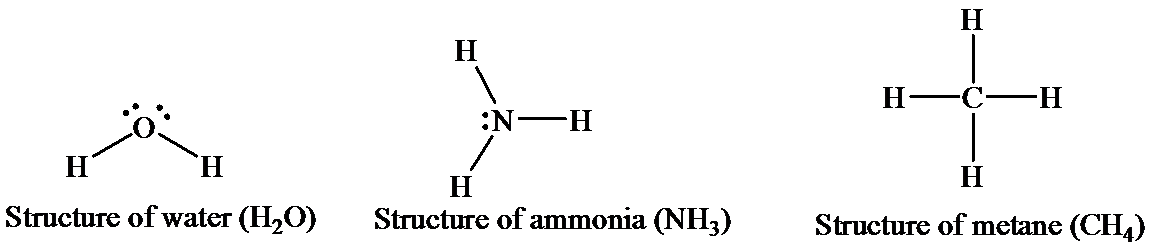
An ammoniamoleculehasonelone pair (unshared electron) ofa nitrogenatom and has a trigonal pyramidal structure. There is limited hydrogen bonding (intramolecular)in case of ammoniaasnitrogen has onlyone lone pair available.
Methane exists in a gaseous statethat has one carbon atom bonded with four hydrogen atoms. The central atom, that is, carbon, forms covalent bonds with four hydrogen atoms by the sharing of electrons. This sharing completes the outer shell of both the carbon and the hydrogen atoms. The four hydrogen atoms give an overall tetrahedral shape to the molecule. No hydrogen bonds are involved in methane as there is no highly electronegative element to form a bond with hydrogen.
A hydrogen bondhaslower bond strength thana covalent bond, thus, it is weaker. However, when a largenumber of intermolecular bonds are formed, they are quite strong. This explains the differences in the heat of fusion (energy required to melt ice by breaking bonds) for water, ammonia, and methane. Due to a highernumber of hydrogen bonds, physical propertie, such asmelting point, boiling point, heat capacity, the heat of fusion, surface tensio, nnd heat of vaporization, have higher values in wate, rs compared to that of methane and ammonia.


In ammonia, the density of the solid state (ice), if generated, is expected to be more than that of liquid ammonia. This is because the hydrogen bonding between different ammonia molecules will not be as extensive as in case of water insolid state (open cage structure in ice). This is due to limited hydrogen bonding of nitrogen. Thus, as a general case, the solid form of ammonia will beless denser than its liquid form.
Thus, it can be concluded that water, ammonia, and methane molecules are tetrahedral in structure, but differ in their overall geometry and physical properties due to thenumber of lone pairs in them and hydrogen bonding. The heat of fusion is the highest for water moleculesbecauseofstrong intermolecular hydrogen bonds. Also, ammonia ice will not form a cage-like structure (as in water), and thus, its density is expected to be lesser than that of liquid ammonia.
Want to see more full solutions like this?
Chapter 3 Solutions
Biochemistry: The Molecular Basis of Life
- Balance the following (fictional) chemical equations by supplying the correct coefficient. Do not leave any space blank (in other words, write in 1 if you would be leaving it blank): R(OZ)2 - RY2+ Z20 a) ZY+ b) D2 (g) + L2 (g) – DL3 (e)arrow_forwardA compound with empirical formula C2H5O was found in a separate experiment to have a molar mass of approximately 90 g. What is the molecular formula of the compound?arrow_forwardElemental analysis of a compound with molar mass 342.3 g/mol gives the following mass percent composition: C 42.11%, H 6.48%, O 51.41%. Find the molecular formula of the compound. Enter your answer in the space below using the following format: if the molecular formula of a compound containing elements X, Y, and Z is X2YZ3 enter your answer as X2YZ3.arrow_forward
- A student ran the following reaction in the laboratory at 759 K: N2(g) + 3H2(g) = 2NH3(g) When she introduced 3.13×10-2 moles of N2(g) and 6.00x102 moles of H2(g) into a 1.00 liter container, she found the equilibrium concentration of NH3(g) to be 6.84×10-4 M. Calculate the equilibrium constant, K., she obtained for this reaction. K. =arrow_forwardThe atomic emission spectrum for a particular element includes blue-violet light with wavelength 440. nm. Calculate the energy in joules of this light given that E = h c/A, and h = 6.63 x 10-34Js, and c = 3.00 x 10°m/s. (h andc are constants, A is wavelength, convert nm into m) h c E =arrow_forwardSolid iron(II) fluoride (FeF2, Ksp= 2.36 x 10-6) is dissolved in water. If 8.1 x 10-3 mol L-1 of iron(II) ion is found to be in solution. Is the solution saturated, unsaturated, desaturated or supersaturated.arrow_forward
- The enthalpy of combustion of hard coal averages −35 kJ/g, that of gasoline, 1.28 × 105 kJ/gal. How many kilograms of hard coal provide the same amount of heat as is available from 1.0 gallon of gasoline? Assume that thedensity of gasoline is 0.692 g/mL (the same as the density of isooctane).arrow_forwardYou are given a solution of 0.117 M NaOH solution. You need to neutralize 10.00 mL of the NaOH solution with an acid called KHP. The reaction is: KHP+ NaOH --> H2O + a salt If you are going to make up a KHP stock solution in a 100.00 mL Volumetric flask and use 14.137 mL of the KHP stock solution to neutralize the NaOH, what mass of KHP would you need to use? The molar mass of KHP is 204.22 g/molearrow_forwardCompound A has molecular formula C7H7X. Its 1H-NMR spectrum shows a singlet at 2.25 ppm and two doublets, one at 7.28 ppm and one at 7.39 ppm. The singlet has an integral of three and the doublets each have an integral of two. The mass spectrum of A shows a peak at m/z = 126 and another peak at m/z = 128; the relative height of the two peaks is 3:1 respectively. Identify what atom X is, explaining your reasoning and identify Compound A, explaining your reasoning.arrow_forward
- For Be-10, find the: a.) mase defect (in grams) b.) binding energy in kilojoules per mole. mass proton= 1.00728 amu; mass neutron= 1.00867 amu; mass Be-10 = 10.013534679 amuarrow_forwardNitrogen reacts with powdered aluminum according to the reaction:2 Al(s) + N2( g) → 2 AlN(s)How many liters of N2 gas, measured at 892 torr and 95 °C,are required to completely react with 18.5 g of Al?arrow_forwardWhat mass of gallium oxide, Ga2O3, can be prepared from 29.0 g of gallium metal? The equation for the reaction is 4Ga + 3O2 ⟶ 2Ga2 O3.arrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
