
(a)
Interpretation: Bonding and anti-bonding molecular orbitals has to be determined for the molecular orbitals of
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
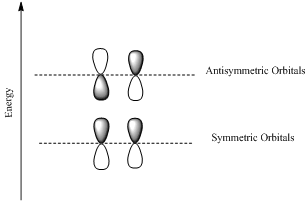
(b)
Interpretation: HOMO and LUMO molecular orbitals in the ground state has to be determined for the molecular orbitals of
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
Ground state and and exited states are the positions with lower and higher energy respectively.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
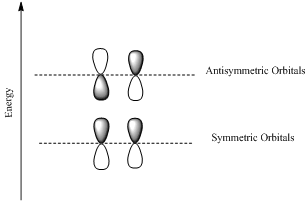
(c)
Interpretation: HOMO and LUMO molecular orbitals in the existed state has to be determined for the molecular orbitals of
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
Ground state and and exited states are the positions with lower and higher energy respectively.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
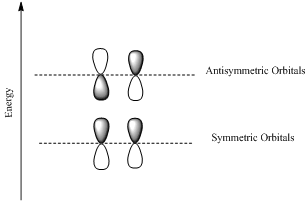
(d)
Interpretation: Symmetric and anti-symmetric MOs has to be determined for the molecular orbitals of
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
Ground state and and exited states are the positions with lower and higher energy respectively.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
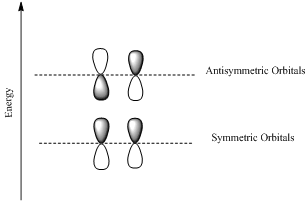
(e)
Interpretation: The relationship between HOMO and LUMO and symmetric and antisymmetric molecular orbitals has to be determined.
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
Ground state and and exited states are the positions with lower and higher energy respectively.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
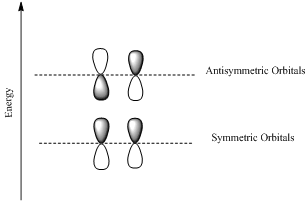
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry (8th Edition)
- Answer the following questions for the molecular orbitals (MOs) of 1,3,5,7-octatetraene: a. How many MOs does the compound have? b. Which are the bonding MOs, and which are the antibonding MOs? c. Which MOs are symmetric, and which are antisymmetric? d. Which MO is the HOMO and which is the LUMO in the ground state? e. Which MO is the HOMO and which is the LUMO in the excited state? f. What is the relationship between HOMO and LUMO and symmetric and antisymmetric orbitals? g. How many nodes does the highest-energy MO of 1,3,5,7-octatetraene have between the nuclei?arrow_forwardThe most stable MO of 1,3,5-hexatriene and the most stable MO of benzene Which compound is more stable? Why?arrow_forward4. The molecular orbital (MO) energy diagram for cyclopentadiene (compound pictured right) shows four distinct pi molecular orbitals at different energy levels. For this question, you do not need to draw all four pi molecular orbitals. Draw the orbital picture of two MOs - the HOMO (highest occupied MO) and the LUMO (lowest unoccupied MO). Label each clearly as HOMO and LUMO, label any "nodes," and shade the orbitals to show in-phase or out-of-phase. maips s benovel ed bluow yswrngarrow_forward
- 1. Which MO of ethylene will it use to accept the metal d-electrons through backbonding? a.σ b.π* c.σ* d.πarrow_forwardAnswer the following questions for 1,3,5- hexatriene, the conjugated triene containing six carbons.Which MOs are bonding? Which are antibonding?arrow_forward2. Which of the following molccules is achiral? HOH HO,CH H. CH, II A)I IV C) II D) IV and Varrow_forward
- 29. Recalling that the orthonitrate anion (NO43) is best described as acyclic, how many non-bonded (lone) electron pairs are there (in total) in this ion in its most important resonance structure? A. 3 non-bonded (lone) electron pairs in total. B. 9 non-bonded (lone) electron pairs in total C. 11 non-bonded (lone) electron pairs in total D. 12 non-bonded (lone) electron pairs in total E. None of the above 30. Given that the orthonitrate anion (NO43) is best described as acyclic, how many resonance structures contribute to its resonance hybrid when using the Octet Rule? A. There is only one (1) important resonance structure B. There are two (2) important resonance structures C. There are three (3) important resonance structures D. There are four (4) important resonance structures E. None of these are correctarrow_forward18. In the molecular orbital representation of cyclobuta-1,3-diene, how many molecular orbitals are present? A) 1 B) 2 C) 4 D) 6 E) 8 and a chatarrow_forwardAnswer the following question for 1,3,5- hexatriene, the conjugated triene containing six carbons. Which MOs are the frontier molecular orbitals?arrow_forward
- 7. Provide a MO diagram, which depicts the relative energies of the pi molecular orbitals of Cyclooctatetraene. Label each orbital. Label the HOMO and the LUMO. Show which MOs are filled in Cyclooctatetraene's ground state (A diagram will be sufficient; you do not have to draw the MOS).arrow_forwardHow many MOs are Bonding MOs in benzene ?arrow_forward5. Molecular Orbitals 2. The carbon-oxygen single bond of carbonyls can be constructed using a mix of both hybridization and MO theory. Construct a MO diagram of the o bond of a carbonyl by mixing an oxygen sp² orbital and a carbon sp² orbital. Make sure your diagram is complete with all necessary orbitals (drawings of them), orbital notations, the proper number of electrons, and an appropriate energy ranking. H3Carrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

