
Concept explainers
Interpretation:
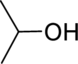
The given below compound has to be matched with the appropriate spectrum.
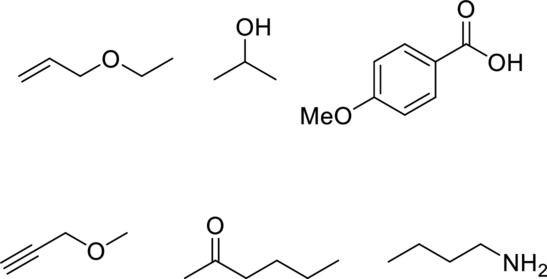
Concept Introduction:
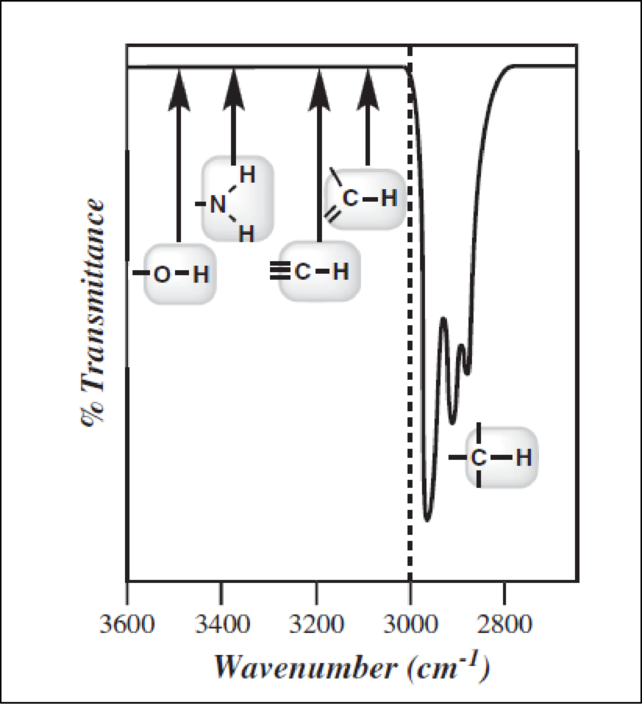
To analyze an IR spectrum certain tools are needed. The first step is to draw a line at
Double bonds:
Triple bonds:
While looking for
Explanation of Solution
Consider spectrum A.
There are no signals in the triple-bond region, but there is one signal in the double-bond region. The signal at
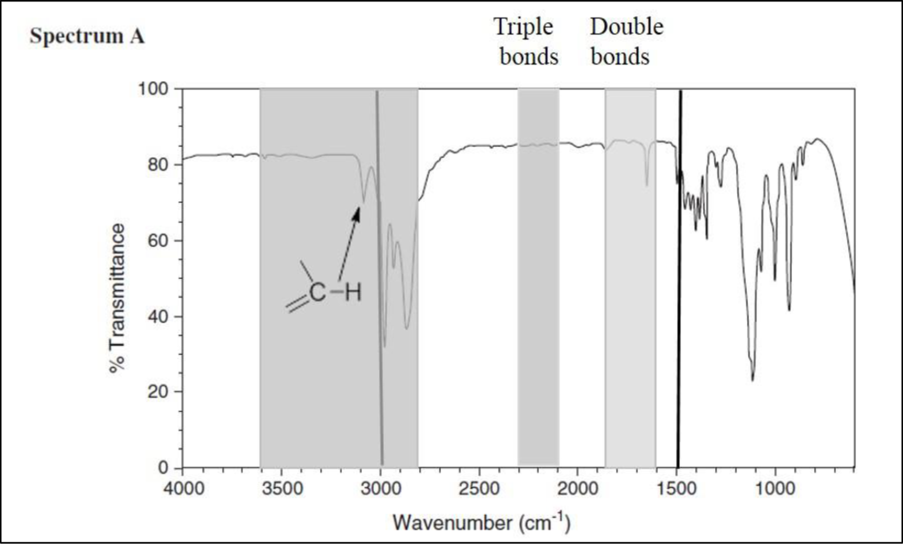
This spectrum exhibits one signal just above
Consider spectrum B.
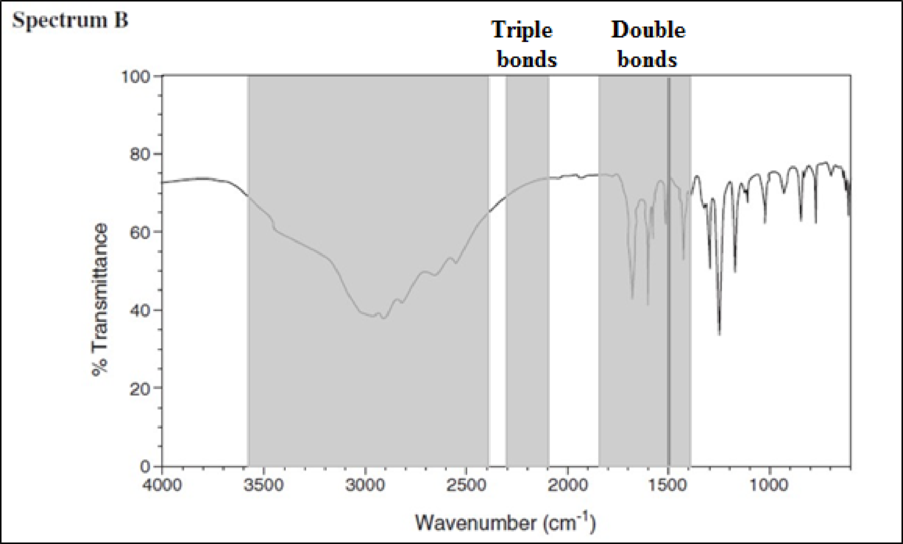
There are no signals in the triple-bond region, but there are signals in the double-bond region. The signals between
Consider spectrum C.
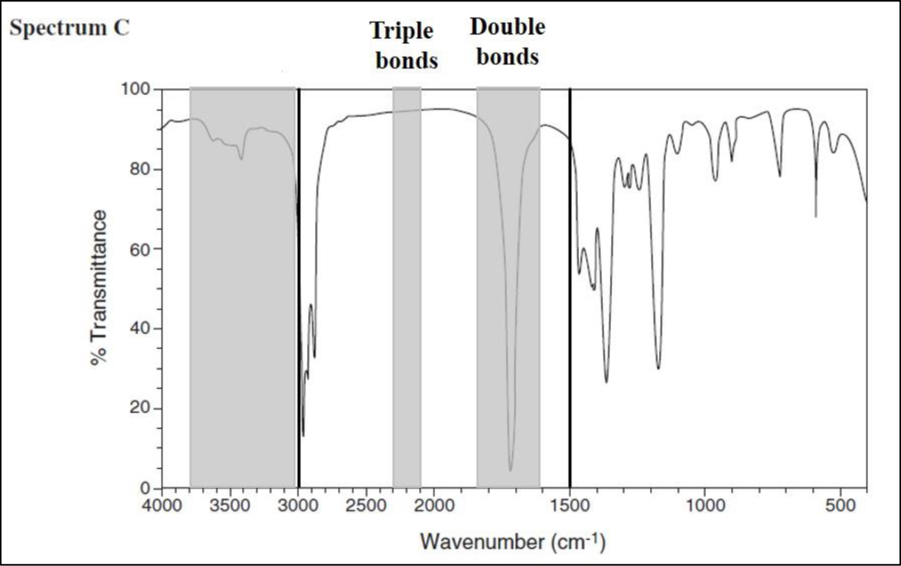
There are no signals in the triple-bond region, but there are signals in the double-bond region. The signal at
The little bump between
Consider spectrum D.
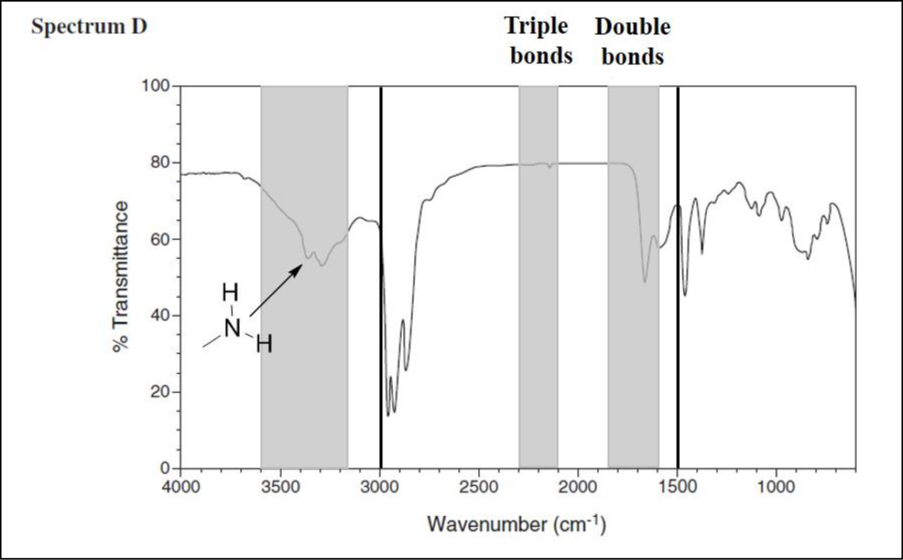
There are no signals in the triple-bond region, but there are signals in the double-bond region. The signal at

Consider spectrum E.
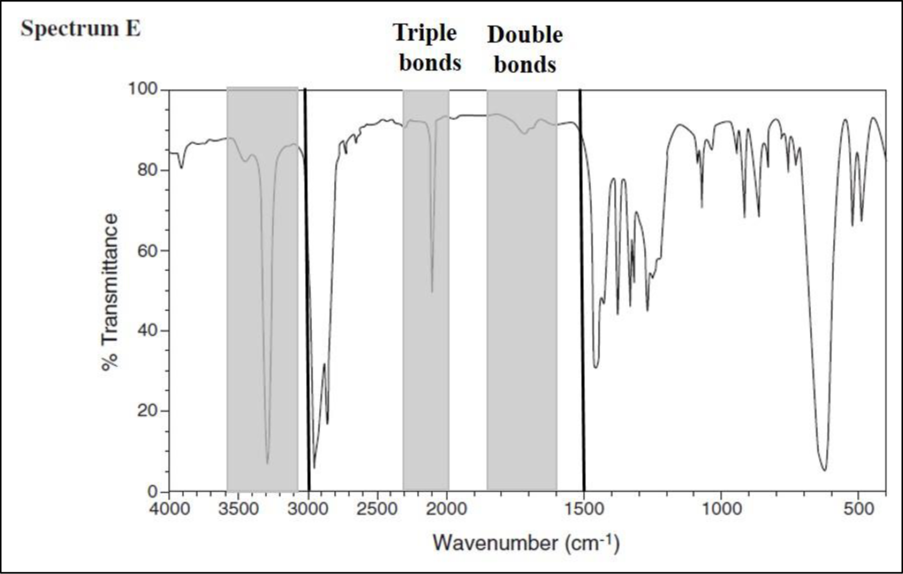
There is a signal in the triple-bond region, but there are no signals in the double-bond region. The signal at

Consider spectrum F.
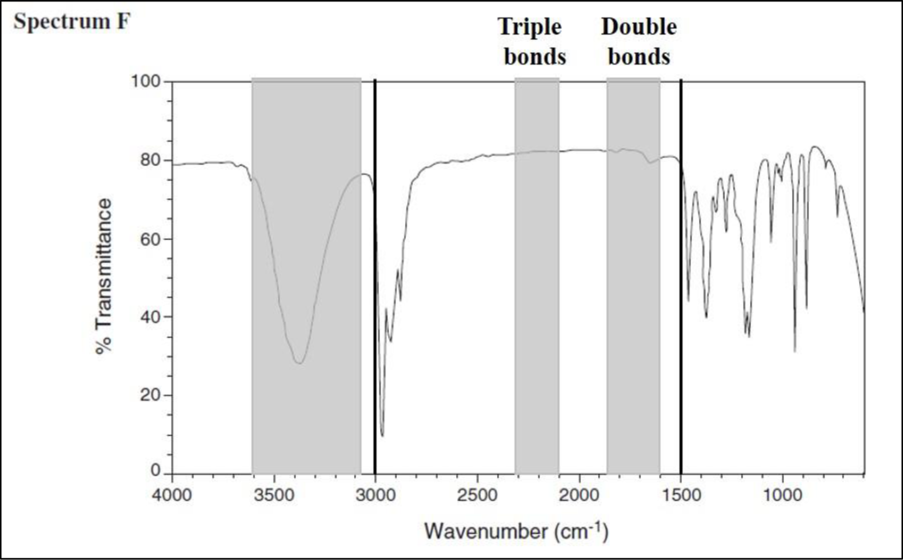
There are no signals in the triple-bond region and also in the double-bond region. Draw a line at
Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
- Draw the structure of the correct compound on each spectrum and match each type of hydrogen on the structure to the corresponding peak on the spectrum. Thanksarrow_forwardEach line in a 13CNMR spectrum corresponds to a different kind of carbon atom. How many 13CNMR signals does each compound exhibit? ( please explain)arrow_forwardChoose the molecule to which each of these two IR spectrums correspond (one possible answer per IR spectrum).arrow_forward
- Match the peaks in this spectrum with hydrogens on the structure below. A) H3C I H₂ 3.0 H₂ CH3 IV 2.0 ppm 1.0 1.68 ppm II: 0.99 ppm III: 2.32 ppm IV: 3.61 ppm 1: B) I: 0.99 ppm II: 1.68 ppm III: 2.32 ppm IV: 3.61 ppm C) I: II: 1.68 ppm 0.99 ppm +arrow_forwardThe 1H-NMR spectrum of 1-chloropropane shows three signals and the 1H-NMR spectrum of 2-chloropropane shows two signals. Draw these two molecules and determine the relative integrals of each signal.arrow_forwardThe structure of citronellal is shown below, with the carbon atoms numbered. Also shown is a table of 13C NMR signals for the molecule and the 13C-NMR DEPT spectrum. Assign each signal in the 13C-NMR spectrum to a numbered carbon in the molecule.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
