
a)
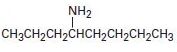
Interpretation:
To explain the Hofmann elimination of the given
Concept introduction:
The Hofmann elimination is an E2 reaction that converts an amine into an
b)
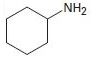
Interpretation:
To explain the Hofmann elimination of the given amines.
Concept introduction:
Just like alcohol, amines can be converted into alkenes by an elimination reaction.
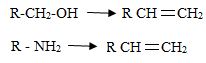
But because an amide ion, -NH2 is a poor leaving group, it must be converted into a better leaving group.
c)
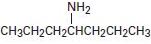
Interpretation:
To explain the Hofmann elimination of the given amines.
Concept introduction:
The given amine has two symmetrical groups is CH3CH2CH2 – attached to the carbon C-4- carrying the amine –NH2 group.
d)
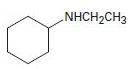
Interpretation:
To explain the Hofmann elimination of the given amines.
Concept introduction:
The Hofmann elimination is an E2 elimination that converts an amine to an alkene and occurs with non-Zaitsev regiochemistry to form the less highly substituted double bond alkene as the major product.
Trending nowThis is a popular solution!

Chapter 24 Solutions
Organic Chemistry
- 2262 Sat Aor 6 1 Which one of the following compounds is the strongest acid? CNH2 -NH2 -CO2H OH CI- СОН CH CH C A B 2. Which one of the following amines will not produce an amide when mixed with an acid chloride? NH PHCH,NH2 PHNH2 Et,NH A C 3. What is the major product of the following sequence of reactions? CH3 1) NBS, hv (excess) C(OH)3 CHO CO2H CH2O 2) NaOH, H20 (excess) A В C 4. What is the major product of the following intramolecular aldol condensation reaction? E NaOH H20 (dehydration) A C 3. D. Barrow_forwardProvide the steps to convert the following aldehyde to the given amine product.arrow_forwardShow how to synthesize the following amines from the indicated starting materials below. Each of the final products may require several sequential steps. Draw the structure for each of the steps and show the reagents required. Starting material Final product b) Benzene m-bromoaniline c) ethylbenzene p-ethylaniline + o-ethylaniline d) benzene p-bromoaniline + o-bromoanilinearrow_forward
- 3. Which of the following amines will not undergo an acyl substitution reaction with methyl acetate (CH3CO2CH3)? A) NH3 B) H₂N C) H₂N D) H3C-N-CH3 CH3 E) All of these will react with methyl acetatearrow_forward1. Which of the following amines would make an imine if reacted with an aldehyde or ketone? A B e. Which of the following moleculo C NH₂ AN Darrow_forward1. Rank the following amines A-D in order of decreasing basicity, where 1 is the most basic. NO2 A B :ZIarrow_forward
- What carboxylic acid and amine are needed to synthesize the pain reliever phenacetin? Phenacetin was once a component of the over-the-counter pain reliever APC (aspirin, phenacetin, caffeine), but it is no longer used because of its kidney toxicity.arrow_forwardGive the major product for the reaction. Pyrrolidine is a secondary amine. CH CH O CH CH! O OH CH2 O CH. I CH.CH Ї TCH CHE OH CH CH₂CH₂ CH.CH CH.OH2 0= RO pyrrolidine. 2 GH CH2 3 HCI H,0 TOH OH: CH₂CH₂ TCH CH: CH₂CH. CH₂CH₂arrow_forwardMethamphetamine is a long-lasting, potent stimulant sold as a street drug. The synthesis is quite simple and one step in the synthesis is shown below. Using your knowledge of the reactions of amines, provide all the reagents necessary to complete the reaction below. Provide the reagents for step 1 and step 2 ,arrow_forward
- Propose a structure for an amine with molecular formula CGH7N by interpreting the given 'H and 13C NMR data below. 1H NMR O 2.35 ppm O7.10 ppm 8 8.46 ppm singlet (3) doublet (2) doublet (2) 13C NMR 2, 4 1,5 6.00- 5.00- 4.00- 3.00- 2.00- 1.00- 0.00- 200 120 100 8/ ppm 180 160 140 80 60 40 20arrow_forwardWhat products are obtained when the following tertiary amines react with hydrogen peroxide followed by heat?arrow_forwardWrite the mechanism for the following reactions: 1. the acid-catalyzed hydrolysis of an imine to a carbonyl compound and a primary amine 2. the acid-catalyzed hydrolysis of an enamine to a carbonyl compound and a secondary amine a. How do the two mechanisms differ?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


