
Concept explainers
a)
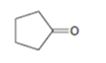
Interpretation:
The enone product expected from the aldol condensation of cyclopentanone is to be given.
Concept introduction:
To give:
The enone product expected from the aldol condensation of cyclopentanone.
b)
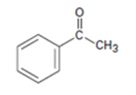
Interpretation:
The enone product expected from the aldol condensation of acetophenone is to be given.
Concept introduction:
Aldehydes and ketones with α-hydrogen undergo a base catalyzed carbonyl condensation reaction in aldol condensation. In this reaction two molecules of the reactant combine by forming a bond between α-carbon of one molecule and the carbonyl carbon of the second molecule. The product obtained, a β-hydroxyaldehyde or ketone upon heating eliminate a molecule of water to yield the enones, α,β-unsaturated aldehydes or ketones.
To give:
The enone product expected from the aldol condensation of acetophenone.
c)
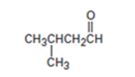
Interpretation:
The enone product expected from the aldol condensation of 2-methylbutanal is to be given.
Concept introduction:
Aldehydes and ketones with α-hydrogen undergo a base catalyzed carbonyl condensation reaction in aldol condensation. In this reaction two molecules of the reactant combine by forming a bond between α-carbon of one molecule and the carbonyl carbon of the second molecule. The product obtained, a β-hydroxyaldehyde or ketone upon heating eliminate a molecule of water to yield the enones, α,β-unsaturated aldehydes or ketones.
To give:
The enone product expected from the aldol condensation of 2-methylbutanal.
Trending nowThis is a popular solution!

Chapter 23 Solutions
Organic Chemistry
- What aldol product is formed when two molecules of butanal react together in the presence of base? What reagents are needed to convert this product to each of the attached compounds?arrow_forwardIn theory, the intramolecular aldol reaction of 6-oxoheptanal could yield the three compounds shown. It turns out, though, that 1-acetylcyclopentene is by far the major product. Why are the other two compounds formed in only minor amounts? Draw a stepwise mechanism to show how all three products are formed.arrow_forwardWhat aldol product is formed when two molecules of butanal react together in the presence of base? What reagents are needed to convert this product to each of the attached compound?arrow_forward
- 1. Each of the following compounds can be prepared by a mixed aldol condensation reaction. Give the structures of the aldehyde and/or ketone precursors for each aldol product shown. soarrow_forwardA different way of synthesising the product 4,4- diphenylbut-3-en-2-one is via an aldol condensation. What starting materials would you use and what reaction conditions would you employ? Draw the full reactionarrow_forwardEach of the following can be prepared by an intramolecular aldol condensation of a diketone. Apply retrosynthetic analysis to deduce the structure of the diketone in each case.arrow_forward
- 16. Draw the structure of the aldol self-condensation (followed by dehydration) product for each of the following compounds. If a compound does not undergo aldol self-condensation, explain why it does not. a) || CH3CHCH₂CH T CH₂ b)arrow_forwardprovide the major product of the crossed aldol reactionsarrow_forwardWhat are the products of an aldol cyclization for the following compound?arrow_forward
- What product would you expect from aldol cyclization of hexanediol in basic solution?arrow_forwardCan 2,4-pentanedione undergo an intramolecular aldol addition? If so, why? If not, why not?arrow_forwardWhich one of the following compound will exhibit aldol condensation reaction towards completion? H H & Harrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

