
Concept explainers
(a)
Interpretation:
The curved arrow mechanism, for base catalyzed exchange of hydrogen
Concept introduction:
A carbonyl group has
Answer to Problem 22.74AP
The curved arrow mechanism for base catalyzed exchange of hydrogen
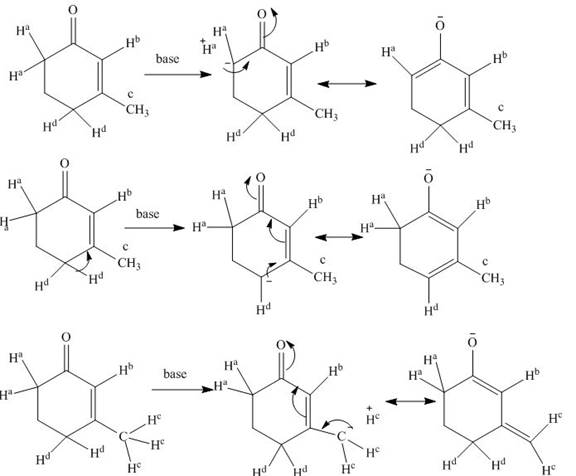
Explanation of Solution
The structure of compound is given below.
Figure 1
When a base is present in the medium, it easily removes the
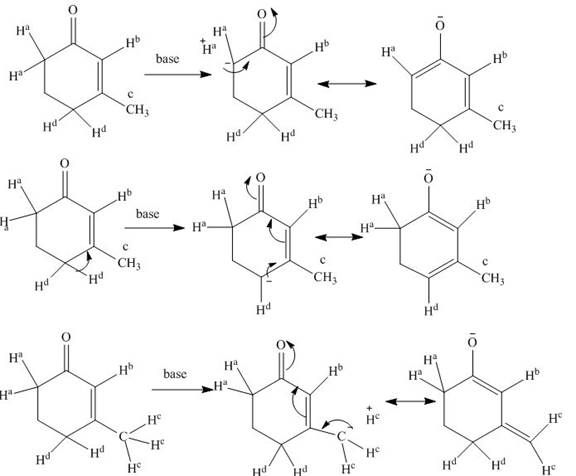
Figure 2
The base catalyzed exchange of hydrogen
(b)
Interpretation:
The less acidic character of
Concept introduction:
The
Answer to Problem 22.74AP
The proton,
Explanation of Solution
The
Figure 3
The
(c)
Interpretation:
The mechanism for exchange of
Concept introduction:
In presence of the acid or base, the enone compounds remain in equilibrium with their isomers. Due to this, acidity of
Answer to Problem 22.74AP
The isomer
Explanation of Solution
The equilibrium of isomers,
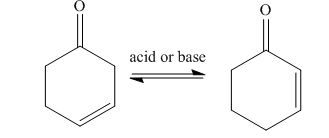
Figure 4
The
The reaction mechanism is shown in figure below.
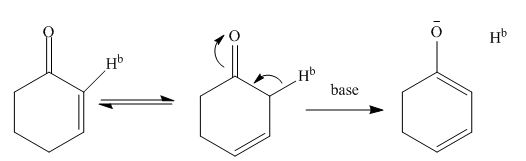
Figure 5
The
(d)
Interpretation:
The hydrogen that is replaced by deuterium in sex hormone, testosterone is to be identified.
Concept introduction:
The
Answer to Problem 22.74AP
The hydrogen at position “a” in vicinity to carbonyl carbon can be exchanged for deuterium in
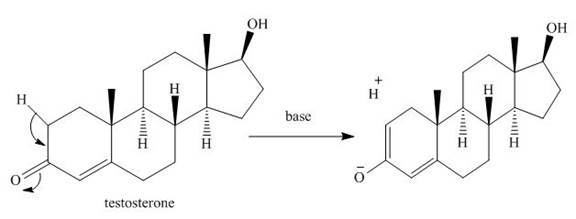
Explanation of Solution
The structure of sex hormone testosterone is given below.
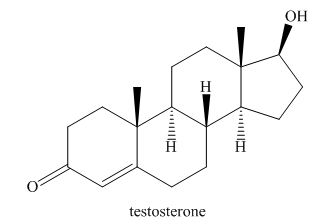
Figure 6
The hydrogen present at position “a” can be exchanged with deuterium, in sex hormone testosterone. This hydrogen, being at
The mechanism for the same can be shown as given below.

Figure 7
The
Want to see more full solutions like this?
Chapter 22 Solutions
Organic Chemistry
- (a) Explain how NaBH, in CH;OH can reduce hemiacetal A to 1,4-butanediol (HOCH,CH,CH,CH,OH). (b) What product is formed when A is treated with Ph;P=CHCH,CH(CH),? (c) The drug isotretinoin is formed by reaction of X and Y. What is the structure of isotretinoin? Although isotretinoin (trade name Accutane or Roaccutane) is used for the treatment of severe acne, it is dispensed under strict controls because it also causes birth defects. PPha NaOCH,CH3 HO- isotretinoin HO A Br X Yarrow_forwardRank the following compounds in order of increasing acidity. CН:CH2CH3, CICH:CH2OH, CHЗCH2OH CН:CH2NH2, (CHз)зN, CH3CH2OН СН:CH-CH3, СНЗСН2ОН, СНЗСH2NH2 а. С.arrow_forwardAlthough p-hydroxybenzoic acid is less acidic than benzoic acid, o-hydroxybenzoic acid is slightly more acidic than benzoic acid. Explain this result. OH но -COOH -соон p-hydroxybenzoic acid o-hydroxybenzoic acidarrow_forward
- Rank the following compounds in order of increasing acidity (1 = least acidic, 3 = most acidic) and in the space provided use resonance (of the conjugate base) to explain why the compound you have labelled “3” is the most acidic.arrow_forward2. (a) MeO Least basic List the following compounds in order of increasing basicity. 265 NH₂ NH₂ NH →>> Most basic NH₂arrow_forwardh) Which is a weaker base and why? Pyridine Pyrrole i) Name the following compounds. H2N A B NH2arrow_forward
- Arrange the following molecules in increasing order of acidity. Base it only on their structural differences and explain how it is so. 1. HF, CH3CH2CH2OH, CH3CH2COOH 2. Ethyl amine, Ethanol, Propanearrow_forwardOf the following carboxylic acids, which is the most acidic? (A) CH3CO₂H (B) HCO₂H (C) CO₂H (D) Cl₂CCO₂H (E) (CH3)3CCO₂Harrow_forwardSection 1: (a) Circle the compound which undergoes decarboxylation when heated. HO2C HO,C CO,H B (b) Which carbon atom has the most acidic hydrogen in the following compound? 2arrow_forward
- 4 5 Arrange the following bases in increasing base strength. There is no partial credit on this problem. Weakest base 1 2 3 Pyridine (C5H5N), Kb = 1.7 x 10-⁹ Aniline (C6H5NH2), Kb = 3.9 x 10-10 Strongest base Methylamine (CH3NH₂), Kb = 4.4 x 10-4 Hydrazine (N2H4), Kb = 1.3 x 10-6 ⠀ Dimethylamine ((CH3)2NH), K₂ = 5.1 x 10-4arrow_forwardPQ-14. What is the major product of this reaction? 1) H₂N-NH2, H3O+ 2) NaOH, 200 °C N-NH2 NHANH2 (B) (D) (A) NHANH2arrow_forwardThe C-H bond in acetone, (CH3)C=O, has a pką of 19.2. Draw two resonance structures for its conjugate base. Then, explain why acetone is much more acidic than propane, CH;CH,CH3 (pKa = 50). %3Darrow_forward
