
Concept explainers
Suggest a reasonable explanation for each of the following observations:
The second-order rate constant
The second-order rate constant for saponification of ethyl
The second-order rate constant
The second-order rate constant
The second-order rate constant
The second-order rate constant
Interpretation:
The reasonable explanation for given observations is to be stated.
Concept introduction:
The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction.
In nucleophilic substitution reaction, nucleophile takes the position of leaving group by attacking on the electron deficient carbon atom.
Answer to Problem 45P
Solution:
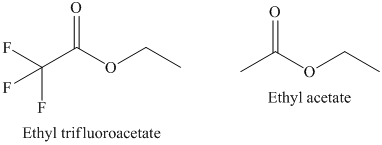
a) The rate constant for saponification of ethyl trifluoroacetate is more than ethyl acetate because the anionic intermediate is stabilized in ethyl trifluoroacetate.
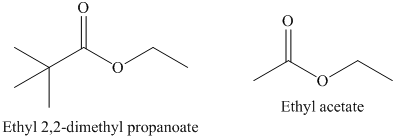
b) The rate constant for saponification of ethyl acetate is more than ethyl
c) The rate constant for saponification of methyl acetate is more than tert-butyl acetate because the incoming nucleophile is less sterically hindered in methyl acetate.
d) The rate constant for saponification of methyl m-nitrobenzoate is more than methyl benzoate because the anionic intermediate is stabilized in methyl m-nitrobenzoate.
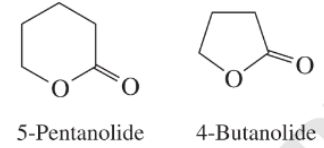
e) The rate constant for saponification of
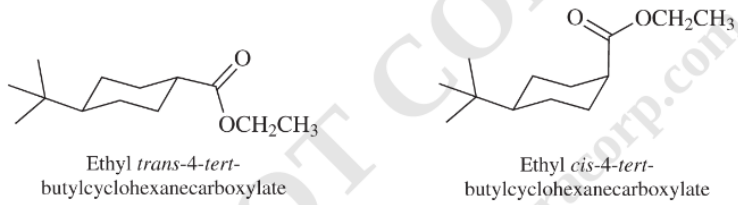
f) The rate constant for saponification of ethyl
Explanation of Solution
a) The saponification of esters involves cleavage of
In ethyl trifluoroacetate,
b) The saponification of esters involves cleavage of
In ethyl acetate,
c) The saponification of esters involves cleavage of
In the first step of saponification reaction, the incoming nucleophile attack on the carbonyl carbon and generate anionic intermediate. In tert-butyl acetate, the attack of incoming nucleophile is sterically hindered by the presence of three methyl groups. However, in methyl acetate only one methyl group is present. Therefore, the second order rate constant for saponification of methyl acetate is much higher than tert-butyl acetate. The structure of methyl acetate and tert-butyl acetate is shown below.
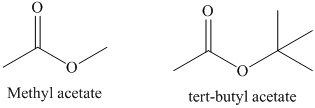
d) The saponification of esters involves cleavage of
In methyl m-nitrobenzoate, nitro group is attached to benzene ring which withdraws the electron density and stabilizes the intermediate. However, in methyl benzoate electron withdrawing group is not attached to benzene ring. It stabilizes anionic intermediate less than the methyl m-nitrobenzoate. Therefore, the second order rate constant for saponification of methyl m-nitrobenzoate is more than methyl benzoate. The structure of methyl m-nitrobenzoate and methyl benzoate is shown below.
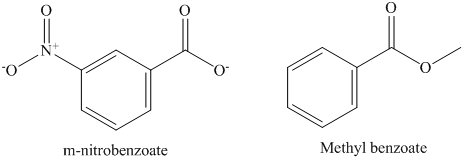
e) The saponification of esters involves cleavage of
The anionic intermediate formed by the saponification of
f) The saponification of esters involves cleavage of
In ethyl
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry - Standalone book
- Calculate the ratio of rates of enzyme-catalyzed to non-catalyzed reactions at 37 °C given that the energy of activation for a particular reaction is reduced from 127 kJ mol−1 to 12 kJ mol−1.arrow_forwardTRUE or FALSE: For a transition state to be considered as the rate- determining step (rds) in a multi-step reaction, it must have the highest change in enthalpy.arrow_forwardThe acid-catalysed conversion of γ-hydroxybutyric acid (GHBA) into its lactone, γ-butyrolactone (GBL) is a reversible reaction. The GHBA to GBL forward reaction is first-order with respect to the GHBA concentration; the GBL to GHBA reverse reaction is first-order with respect to the GBL concentration. An experimental study of the kinetics of this reaction was undertaken in 0.2 mol L-1hydrochloric acid (HCl) at 298 K. The initial concentration of GHBA was 18.23 × 10-3mol L-1 . The concentration of GBL in solution was followed as a function of time (t), as indicated in Table B.3: Time/min 0 21 36 50 65 80 100 ∞ GBL Concentration / 10-3 L-1 0 2.41 3.73 4.96 6.10 7.08 8.11 13.28 Use the data in Table B.3 to determine the equilibrium constant and the first-order rate constants for both forward and reverse reactions.arrow_forward
- There are two molecules with the formula C3H§. Propene, CH;CH= CH2, is the monomer of the polymer polypropylene, which is used for indoor-outdoor carpets. Cyclopropane is used as an anesthetic: CH, CH2 CH, When heated to 499 °C, cyclopropane rearranges (isomerizes) and forms propene with a rate constant of 5.95 x 104s1. what is the half-life of this reaction? What fraction of the cyclopropane remains after 0.75 h at 499.5 °C?arrow_forwardFor the gas phase decomposition of acetic anhydride, (CH3CO) 20→ CH₂=C=O + CH3COOH the rate constant at 502 K is 1.02 x 10³ s1 and the rate constant at 551 K is 0.0219 s¹. The activation energy for the gas phase decomposition of acetic anhydride is kj.arrow_forwardIn aqueous solution buffered at a pH of 6.75 Penicillin G (molecular weight of 334.39 g/mole) undergoes hydrolysis via a pseudo first order reaction that has an activation energy of 77.3 kJ/mole and a pseudo first order rate constant of 1.58 x 10-7 s-1 at 30°C. The solubility of the sodium salt of Penicillin G is 25 mg/ml of water. One would expect the solubility of the sodium salt to be ____________ that of the Penicillin G. a. higher than b. the same as c. lower thanarrow_forward
- A reaction has Keq' = 120. The rate constant of this reaction was found to be 1.11x 101s-1. If the catalytic power of an enzyme for this reaction is 1.25 x 10', what is the rate constant, in s1, for the reverse direction of the catalyzed reaction?arrow_forwardThe enzyme carbonic anhydrase catalyzes the following reaction: CO2 (g) + H2O(1) - HCO3 (aq) +H+(aq) -1 In water, without the enzyme, the reaction proceeds with a rate constant of 0.039 s at 24 °C. In the presence of the enzyme in water, the reaction proceeds with a rate constant of 1.0 ×106 s-1 at 24 °C. Part A Assuming the collision factor is the same for both situations, calculate the difference in activation energies for the uncatalyzed (Ea) versus the enzyme-catalyzed (Eac) reaction. Express your answer in kilojoules to two significant figures. Ea – Eac = kJ Submit Request Answerarrow_forwardThe rate law for the reaction of phenolphthalein with NaOH was determined to be Rate = k[P2−][OH−]. What are the units on the value of the rate constant, k?arrow_forward
- Carbonic Anhydrase or carbonic dehydratase is a family of metalloenzymes containingzinc (Zn2+) ion in its active site. Carbonic anhydrase can greatly increase the rate of thisreaction, reaching a reaction rate of 104 - 106 per second. It is defined as the enzyme found inred blood cells, other parts of animals and plants, that breaks down carbonic acid with carbondioxide and water.CO2(g) + H2O(l) → H2CO3(aq)For the carbonic anhydrase activity specified by the equation above, evaluate the standardreaction Gibbs energy at 25 °C?arrow_forwardIn aqueous solution buffered at a pH of 6.75 Penicillin G (molecular weight of 334.39 g/mole) undergoes hydrolysis via a pseudo first order reaction that has an activation energy of 77.3 kJ/mole and a pseudo first order rate constant of 1.58 x 10-7 s-1 at 30°C. The solubility of the sodium salt of Penicillin G is 25 mg/ml of water. A solution containing the sodium salt of Penicillin G is determined to have a concentration of 1.25 x 10-3 M. The solution is… a. unsaturated b. saturated c. supersaturatedarrow_forwardwrite a rate law for the following reaction if reaction order n=1 2 N2O5 (g) -> 4 NO2 (g) + O2 (g)arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

