
Concept explainers
(a)
Interpretation:
It is to be indicated whether the given conversions would be energetically favorable or unfavourable. Also, it is to be given if the reaction is likely to occur readily.
Concept introduction:
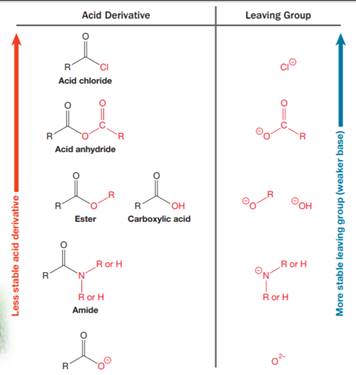
Nucleophilic acyl substitution describes a class of substitution reactions involving nucleophiles and acyl compounds. An acid derivative has a leaving group, which is substituted by a nucleophile. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:
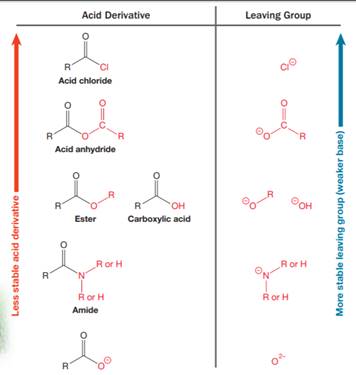
An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Answer to Problem 20.5YT
The given conversion would be energetically unfavourable, and the reaction does not occur readily.
Explanation of Solution
The given reaction is
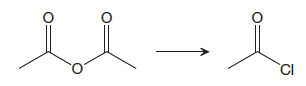
This is an example of a nucleophilic acyl substitution reaction in which an anhydride is getting converted into an acyl chloride. An acid derivative has a leaving group, which is substituted by a nucleophile. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:
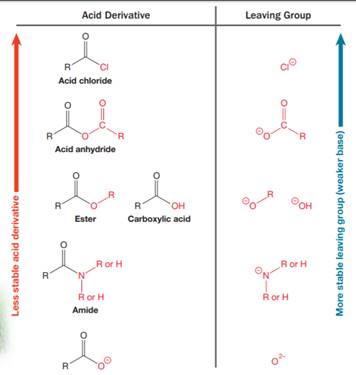
An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to the one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Referring to this chart, an acid chloride is from a lower rung while an acyl chloride is from a higher rung on the stability ladder. Thus, this reaction is energetically unfavorable and so does not occur readily.
Conversion of an acid derivative from a lower rung to higher rung on the stability ladder is energetically unfavourable, and the reaction does not occur readily.
(b)
Interpretation:
It is to be indicated whether the given conversions would be energetically favorable or unfavourable. Also, it is to be given if the reaction is likely to occur readily.
Concept introduction:
Nucleophilic acyl substitution describes a class of substitution reactions involving nucleophiles and acyl compounds. An acid derivative has a leaving group, which is substituted by a nucleophile. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:
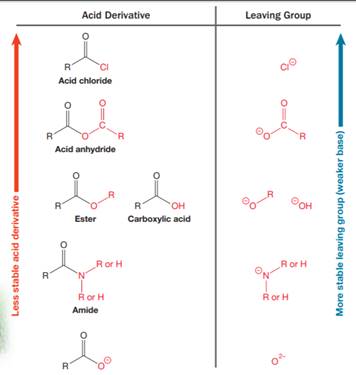
An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Answer to Problem 20.5YT
The given conversion would be energetically unfavourable, and it does not occur readily.
Explanation of Solution
The given reaction is
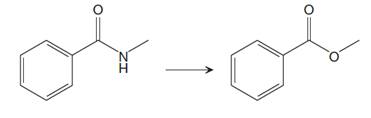
This is an example of a nucleophilic acyl substitution reaction in which an amide is getting converted into an ester. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:
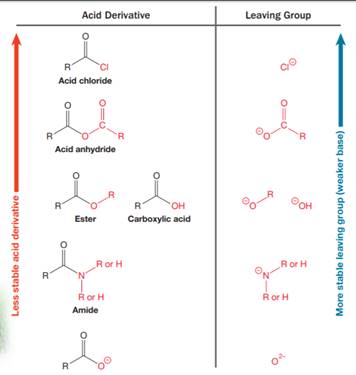
An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Referring to this chart, an amide is from a lower rung while an ester is from a higher rung on the stability ladder. Thus, this reaction is energetically unfavorable and so does not occur readily.
Conversion of an acid derivative from a lower rung to higher rung on the stability ladder is energetically unfavourable, and the reaction does not occur readily.
(c)
Interpretation:
It is to be indicated whether the given conversions would be energetically favorable or unfavourable. Also, it is to be given if the reaction is likely to occur readily.
Concept introduction:
Nucleophilic acyl substitution describes a class of substitution reactions involving nucleophiles and acyl compounds. An acid derivative has a leaving group, which is substituted by a nucleophile. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:
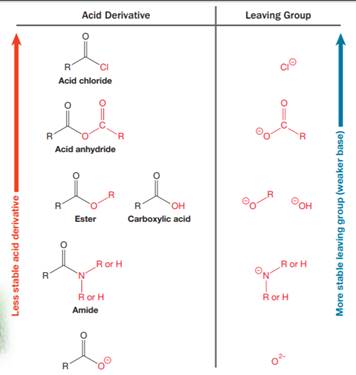
An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Answer to Problem 20.5YT
The given conversion would be energetically unfavourable, and it does not occur readily.
Explanation of Solution
The given reaction is
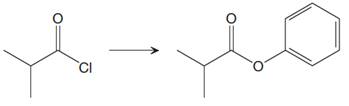
This is an example of nucleophilic acyl substitution reaction in which an acid chloride is getting converted into an ester. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:
An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Referring to this chart, an acid chloride is from a higher rung while an ester is from a lower rung on the stability ladder. Thus, this reaction is energetically favorable and occurs readily.
Conversion of an acid derivative from a higher rung to lower rung on the stability ladder is energetically favourable, and the reaction occurs readily.
(d)
Interpretation:
It is to be indicated whether the given conversions would be energetically favorable or unfavourable. Also, it is to be given if the reaction is likely to occur readily.
Concept introduction:
Nucleophilic acyl substitution describes a class of substitution reactions involving nucleophiles and acyl compounds. An acid derivative has a leaving group, which is substituted by a nucleophile. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:

An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Answer to Problem 20.5YT
The given conversion would be energetically unfavourable, and it does not occur readily.
Explanation of Solution
The given reaction is
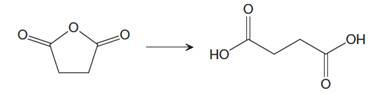
This is an example of a nucleophilic acyl substitution reaction in which an acid anhydride is getting converted into dicarboxylic acid. If the leaving group is more stable than the nucleophile (weaker in terms of basicity), then the reaction is energetically favorable and occurs readily. If the nucleophile is more stable than the leaving group, then the reaction is enegetically unfavorble and does not occur readilty. This can be explained on the basis of the stability ladder below:

An acyl substitution that converts an acid derivative from a higher rung on the stability ladder to one on a lower rung of the ladder is energetically favorable. An acyl substitution that converts an acid derivative from a lower rung on the stability ladder to one on a higher rung of the ladder is energetically unfavorable.
Referring to this chart, an acid anhydride is from a higher rung while a carboxylic acid is from a lower rung on the stability ladder. Thus, this reaction is energetically favorable and occurs readily.
Conversion of an acid derivative from a higher rung to lower rung on the stability ladder is energetically favourable, and the reaction occurs readily.
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Draw the product & reaction arrows for each of the following acid-base reactions for the following. Which side of the equilibrium will be favored in both reactions?arrow_forwardWhich of the following two reactions would you predict to go faster and why?.arrow_forwardWhich of the following will react faster and whyarrow_forward
- Supply the missing arrows to complete the reaction mechanisms below. For each of the reactions, BOX the electrophile and ENCIRCLE the nucleophile.arrow_forwardDraw a complete, step-wise, curved arrow mechanism for each reaction shown below. You don't need to worry about stereochemistry for these problems. It may help if you take the following steps. 1) Find the nucleophile and the electrophile. 2) Determine the major functional group present in the nucleophile and electrophile. 3) Determine the type of reaction this particular nucleophile/electrophile pair is likely to participate in 4) Draw the mechanism that corresponds with this reaction type. a) OH cat. H2SO4 HO Cl2 b) :OHarrow_forwardConsider the reaction between 2-methyl-2-butanol and HBr, shown below. но HBr Brarrow_forward
- The following reaction, which is discussed in Chapter 8, is an example of a unimolecular nucleophilic substitution (Sn1) reaction. It consists of the four elementary steps shown here. For each step (i-iv), (a) identify all electron-rich sites and all electron-poor sites, (b) draw in the appropriate curved arrows to show the bond formation and bond breaking that occur, and (c) name the elementary step. (i) H-OCH3 OH + H-OCH3 H. H (ii) ©CH2 + H,O (iii) ©CH2 CH3 + HO-CH3 (iv) CH3 CH3 + H2O-CH, + НО—СНЗ foarrow_forwardIn (e), please note that the heterocyclic oxygen atoms contains two lone pairs that could contribute to the resonance stabilization of any proposed intermediate.arrow_forwardFor each set of conditions, circle the favored reaction(s) and draw the major product(s). Assume that these reactions are irreversible. If both products are expected to form in comparable yields, circle both reactions and draw both products.arrow_forward
- Draw the structures of the missing reactants, intermediates, or products in the following mechanism. Include all lone pairs.arrow_forwardA proton transfer reaction can occur when an aldehyde is placed in strong base, such as an alkoxide ion, producing an alcohol and a charged conjugate base that is resonance stabilized. In the left box, draw the curved arrows for the proton transfer. In the middle and right boxes, draw the two structures for the resonance-stabilized product as noted in the box-specific directions. Be sure to include all lone pairs and nonzero formal charges.arrow_forwardDetermine the product for each of the following reactions and determing which product will predominate.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
