
Concept explainers
(a)
Interpretation:
The synthesis of the given compound using the cyclopentanone as one of the reagents is to be suggested.
Concept introduction:
In aldol condensation, the
Answer to Problem 18.69P
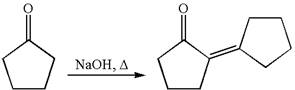
The synthesis of the given compound using cyclopentanone as one of the reagents is:
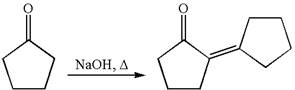
Explanation of Solution
The given compound is:
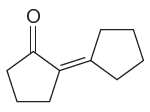
The given compound is a
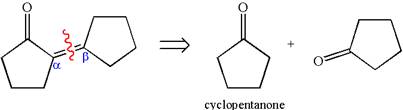
The precursor determined is cyclopentanone. Two molecules of cyclopentanone are used in the aldol condensation. Cyclopentanone, on reaction with a strong base, forms the given
The synthesis of the given compound is planned by identifying the precursor used with disconnection of
(b)
Interpretation:
The synthesis of the given compound using cyclopentanone as one of the reagents is to be suggested.
Concept introduction:
In aldol condensation, the aldehyde or ketone with
Answer to Problem 18.69P
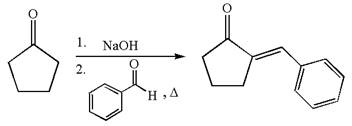
The synthesis of the given compound using cyclopentanone as one of the reagents is:
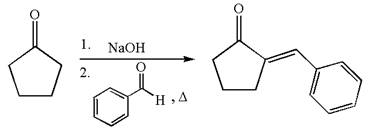
Explanation of Solution
The given compound is:
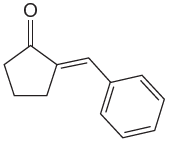
The given compound is
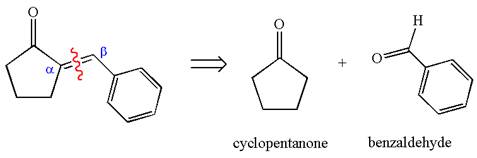
The precursors determined are cyclopentanone and benzaldehyde. As the benzaldehyde has no
The synthesis of the given compound is planned by identifying the precursor used with disconnection of
(c)
Interpretation:
The synthesis of the given compound using cyclopentanone as one of the reagents is to be suggested.
Concept introduction:
In aldol condensation the aldehyde or ketone with
Answer to Problem 18.69P
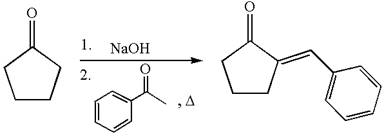
The synthesis of the given compound using cyclopentanone as one of the reagents is:
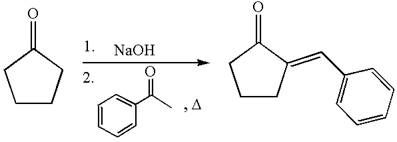
Explanation of Solution
The given compound is:
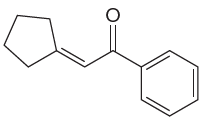
The given compound is
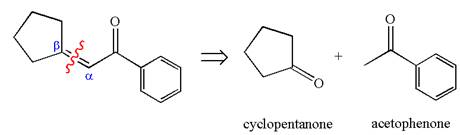
The precursors determined are the cyclopentanone and acetophenone. As the both ketones has
The synthesis of the given compound is planned by identifying the precursor used with disconnection of
(d)
Interpretation:
The synthesis of the given compound using cyclopentanone as one of the reagents is to be suggested.
Concept introduction:
In aldol condensation the aldehyde or ketone with
Instead of carbonyl compounds, the compounds with polar
Answer to Problem 18.69P
The synthesis of the given compound using cyclopentanone as one of the reagents is:
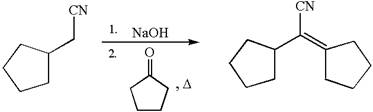
Explanation of Solution
The given compound is:
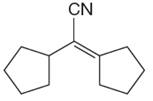
The given compound is
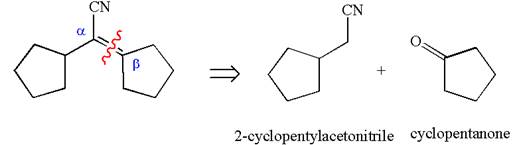
The precursors determined are cyclopentanone and nitrile compound. As the nucleophile adds to carbonyl, the nucleophile can be produced by the reaction of one of the nitrile with a strong base like hydroxide. The nucleophile then reacts with ketone and produces the given
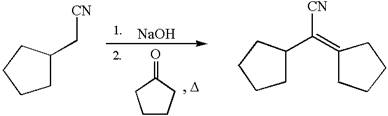
The synthesis of the given compound is planned by identifying the precursor used with disconnection of
(e)
Interpretation:
The synthesis of the given compound using cyclopentanone as one of the reagents is to be suggested.
Concept introduction:
The Robinson annulations reaction is the addition of enolate to conjugated carbonyl followed by intramolecular aldol condensation. One of the reactant in Robinson annulations must be
Answer to Problem 18.69P
The synthesis of given compound using cyclopentanone as one of the reagents is:
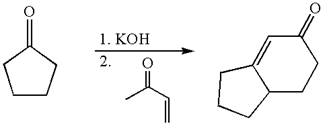
Explanation of Solution
The given compound is:
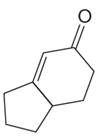
The given compound is
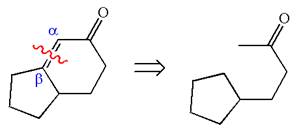
To get the precursor
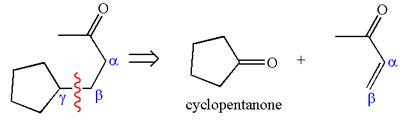
The precursors determined are the cyclopentanone and
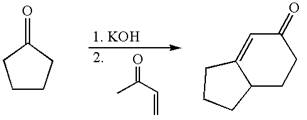
The synthesis of given compound is planned by identifying the precursor used with disconnection of
Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- (SYN) Show how to carry out each of the following syntheses, using any reagents necessary. Hint: In each case, the carbonyl group of a ketone or aldehyde is entirely removed. (a) (b) ? OCH3 OCH3 OCH3 OCH3 (c) (d) ? ? OH Pharrow_forwardIn the boxes provided, predict the major product in each of the following reaction.arrow_forward(SYN) Suggest how you would synthesize each of the following compounds beginning with pentan-2-one. (а) OH OH (b) ОН (c) OHarrow_forward
- A reasonable synthesis for each of the following compoundsarrow_forwardWhich reaction in each of the following pairs would you expect to be faster? (i) Write both reactions using bond-line presentation and, using arrows providing a mechanistic explanation of the course of the reaction, draw either a transition state or an intermediate product of the reaction; (ii) Shortly (one sentence) explain your reasoning why one of the two reactions will be faster.arrow_forward(SYN) Stilbene is used in the manufacture of dyes and also has estrogenic activity. Show how to synthesize stilbene using benzene and any carboxylic acid as your only sources of carbonarrow_forward
- Provide a detailed mechanism for the followingarrow_forward(SYN) Show how each of these compounds can be produced from an alkene or alkyne. Draw the appropriate alkene or alkyne and include any necessary reagents and special reaction conditions. (a) (b) (c) (d) ОН HOarrow_forwardPropose mechanism consistent with the following reaction.arrow_forward
- (SYN) Starting with acetyl chloride (CH3COCl) and any other reagents necessary, how would you synthesize each of the following compounds?arrow_forward(SYN) Show how to carry out each of the following transformations. (a) (b) ? ? (c) (d) ? ? CI HOarrow_forwardThe reaction shown here is an example of the Favorskii reaction, which involves an R¯ leaving group in a nucleophilic addition-elimination reaction. (a) Draw the complete, detailed mechanism for this reaction and explain why R can act as a leaving NaOH H2O group. (b) Suggest how you can synthesize an ester from cyclopropanone using only this reaction.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





