
Concept explainers
Draw the structure corresponding to each name.
a. 3-methyihexanoic acid
b. 3-hydroxy-4-methylheptanoic acid
c. p-nitrobenzoic acid
d. sodium hexanoate
e. m-ethylbenzoic acid
f. propyl decanoate
(a)
Interpretation:
The structure of 3-methylhexanoic acid should be drawn.
Concept Introduction:
The structure of organic compound is drawn in order to represent the arrangement of atoms in which they are present in a molecule.
Answer to Problem 44P
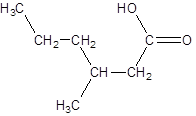
Explanation of Solution
An organic compound in which carboxy functional group that is -COOH is bonded to the carbon atom is said to be a carboxylic acid. The general formula for carboxylic acid is RCOOH or RCO2H.
The IUPAC names are given to the carboxylic acid according to the following steps:
1. The parent (longest) alkane chain is identified.
2. The ending of the parent chain from alkane (-e) is changed to -oic acid for a carboxylic acid group.
3. The numbering is of the chain is done in such a way that carbonyl carbon gets the smaller number.
4. Name should be written in alphabetical order and other substituents are shown by the number.
For number of carbons atoms chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
The given name is 3-methylhexanoic acid where the parent chain is hexane that is 6 carbon atom chain having a methyl substituent at carbon-3. So, the structure of 3-methylhexanoic acid is:
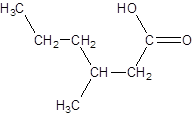
(b)
Interpretation:
The structure of 3-hydroxy-4-methylheptanoic acid should be drawn.
Concept Introduction:
The structure of organic compound is drawn in order to represent the arrangement of atoms in which they are present in a molecule.
Answer to Problem 44P
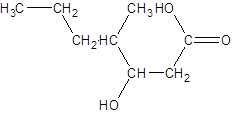
Explanation of Solution
An organic compound in which carboxy functional group that is -COOH is bonded to the carbon atom is said to be a carboxylic acid. The general formula for carboxylic acid is RCOOH or RCO2H.
The IUPAC names are given to the carboxylic acid according to the following steps:
1. The parent (longest) alkane chain is identified.
2. The ending of the parent chain from alkane (-e) is changed to -oic acid for a carboxylic acid group.
3. The numbering is of the chain is done in such a way that carbonyl carbon gets the smaller number.
4. Name should be written in alphabetical order and other substituents are shown by the number.
For number of carbons atoms chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
The given name is 3-hydroxy-4-methylheptanoic acid where the parent chain is heptane that is 7 carbon atom chain having a hydroxy substituent at carbon-3 and a methyl substituent at carbon-4. So, the structure of 3-hydroxy-4-methylheptanoic acid is:
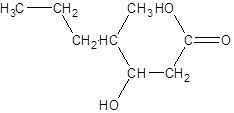
(c)
Interpretation:
The structure of p-nitrobenzoic acid should be drawn.
Concept Introduction:
The structure of organic compound is drawn in order to represent the arrangement of atoms in which they are present in a molecule.
Answer to Problem 44P
Explanation of Solution
An organic compound in which carboxy functional group that is -COOH is bonded to the carbon atom is said to be a carboxylic acid. The general formula for carboxylic acid is RCOOH or RCO2H.
The IUPAC names are given to the carboxylic acid according to the following steps:
1. The parent (longest) alkane chain is identified.
2. The ending of the parent chain from alkane (-e) is changed to -oic acid for a carboxylic acid group.
3. The numbering is of the chain is done in such a way that carbonyl carbon gets the smaller number.
4. Name should be written in alphabetical order and other substituents are shown by the number.
For number of carbons atoms chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
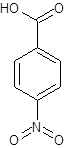
The given name is p-nitrobenzoic acid where the parent chain is benzene having a nitro substituent at carbon-4. So, the structure of p-nitrobenzoic acid is:
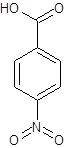
(d)
Interpretation:
The structure of sodium hexanoate should be drawn.
Concept Introduction:
The structure of organic compound is drawn in order to represent the arrangement of atoms in which they are present in a molecule.
Answer to Problem 44P
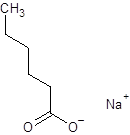
Explanation of Solution
An organic compound in which carboxy functional group that is -COOH is bonded to the carbon atom is said to be a carboxylic acid. The general formula for carboxylic acid is RCOOH or RCO2H. When H of hydroxyl group present in carboxylic acid is replaced by an atom then it results in the formation of respective salt.
The IUPAC names are given to the carboxylic acid salt according to the following steps:
- The parent (longest) carbon chain is identified.
- The name of metal is written first from which the salt is made up of.
- The ending of the for a carboxylic acid group is changed to -oate for naming salt of carboxylic acid.
- The numbering is of the chain is done in such a way that carbonyl carbon gets the smaller number.
- Name should be written in alphabetical order and other substituents are shown by the number.
For number of carbons atoms chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
The given name is sodium hexanoate where the parent chain is hexane having 6 carbon atoms and metal is sodium. So, the structure of sodium hexanoate is:
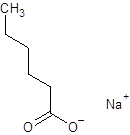
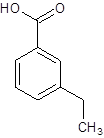
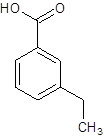
(e)
Interpretation:
The structure of m-ethylbenzoic acid should be drawn.
Concept Introduction:
The structure of organic compound is drawn in order to represent the arrangement of atoms in which they are present in a molecule.
Answer to Problem 44P
Explanation of Solution
An organic compound in which carboxy functional group that is -COOH is bonded to the carbon atom is said to be a carboxylic acid. The general formula for carboxylic acid is RCOOH or RCO2H.
The IUPAC names are given to the carboxylic acid according to the following steps:
1. The parent (longest) alkane chain is identified.
2. The ending of the parent chain from alkane (-e) is changed to -oic acid for a carboxylic acid group.
3. The numbering is of the chain is done in such a way that carbonyl carbon gets the smaller number.
4. Name should be written in alphabetical order and other substituents are shown by the number.
For number of carbons atoms chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
The given name is m-ethylbenzoic acid where the parent chain is benzene having an ethyl substituent at carbon-3. So, the structure of m-ethylbenzoic acid is:
(f)
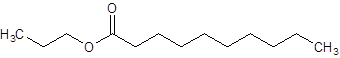
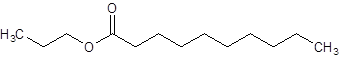
Interpretation:
The structure of propyl decanoate should be drawn.
Concept Introduction:
The structure of organic compound is drawn in order to represent the arrangement of atoms in which they are present in a molecule.
Answer to Problem 44P
Explanation of Solution
An organic compound in which carboxy functional group that is -COOH is bonded to the carbon atom is said to be a carboxylic acid. The general formula for carboxylic acid is RCOOH or RCO2H. When -H of the carboxylic acid is replaced by an alkyl or aryl group (-R') then it results in the formation of an ester having general formula RCOOR'.
The reaction which results in the formation of at least one ester along with water on heating acids with alcohols is said to be esterification.
So, in order to give the IUPAC name to the esters, the following steps are followed:
- The alkyl substituent from the alcohol is named first.
- The name of the parent chain from carboxylic acid part is replaced as carboxylate.
In order to write the common name of the esters, the common of acids are written from which the ester has been formed.
The given name is propyl decanoate where propyl name is derived from propanol and decanoate is derived from the name decanoic acid. So, the structure of propyl decanoate is:
Want to see more full solutions like this?
Chapter 17 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Draw the structure corresponding to each name. a. 1-decanamine b. tricyclohexylamine c. p-bromoaniline d. 3-aminobutanoic acid e. N,N-dipropyl-2-octanamine f. N-ethylhexylaminearrow_forwardон C-H ÓR Compound that contains a group like that shown above. a. acetal Ob. aldehyde c. Benedict's reagent d. carbonyl Oe. hemiacetal Of. hemiketal Og. hydrolysis h. ketal i. ketone Oj. Tollen's reagent 2.arrow_forward6. Draw the structure of the following compounds. a. cyclobutanecarboxylic acid b. 3,3-dimethylpentanedioic acid c. 4-aminopentanoic acid d. 2-methylcyclohexanecarboxylic acid e. m-chlorobenzoic acidarrow_forward
- 6. Draw the following compounds. a. 2-pentanol b. ethylpropylamine c. methyl propyl ether d. butyl propyl ketone abayoqtheo ghidollo WHO H3--3 HO HO HO HO HOarrow_forwardReduction of an alkyl azide results in the formation of —-. A. an imine B. an oxime C. a tertiary amine D. a secondary amine E. a primary aminearrow_forwardCyclopentanone is treated with chlorine (Cl2) in the presence of acid (H*). What product is formed? A. Chlorocyclopentane B. 1-chloro-1-hydroxycyclopentane C. 2-chlorocyclopentanone D. 3-chlorocyclopentanonearrow_forward
- Name : R NH2 Select one: a. alcohols b. amine c. phenols d. aldehydearrow_forwardWhat is the name of the major product formed during the reaction between berzoyl chloride and phenol? a. phenyl benzoate b. benzyl ester C. cyclopentanoate d. benzyl phenoate e. benzenecarboxylic acid O a O b O c earrow_forwardFLAVORANTS: Identify the aldehyde and ketone-containing flavoring compound from each following sources. Choose your best answer from the choices below A. 2-Octanone B. Citral C. Benzaldehyde D. Vanillin E. Cinnamaldehyde F. a-Damascone 1. Вerry 2. Mushroom 3. Lemongrass 4. Cinnamon 5. Almonds Idchyde or ketone formed froarrow_forward
- Which is propyl propanoate? A. CH₂CH₂CH₂OOCCH₂CH; B. CH₂CH₂CH₂COOCH₂CH₂ C. CH₂CH₂CH₂COCH₂CH₂ D. CHỊCH,CH,OCH,CHỊCH, A B C Darrow_forward1. An alkene reacts with water with an acid catalyst results into a formation of: A. Aldehyde B. Ketone C. Alcohol D. Ester 2. 3-Methylhexanal with K2Cr2O7 will yield: A. 3-Methyl-1-hexanol B. 3-Methylhexanoic acid C. 3-Methyl-1-hexanone D. 3-Methyl-1-hexanethiol 3. This is a reverse process of Hydration reaction: A. Oxidation reaction B. Reduction reaction C. Dehydration reaction D. Hydration reaction 4. Acetic acid reacts with a strong base forms: A. Salt B. Water C. Salt and Water D. No reaction 5. Ketones can be further oxidized with benedict's solution into: A. Alcohol B. Aldehyde C. Catalysts D. No reactionarrow_forwardArrange the compounds of each group in order of increasing boiling point. a. 1-butanol, 1-pentanol, 1-propanol b. 1,2-butanediol, 2-butanol, pentanearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





