
Interpretation:
The principal organic products formed in each of the given reactions are to be identified.
Concept Introduction:
In the
The naming of chiral center and geometric isomers are based on Cahn-Ingold-Prelog priority rules. According to these rules, if the priority assigned to each group attached to the chirality center in a molecule is in a clockwise direction, then it is the R-stereoisomer.
Tosylate is a good leaving group. Tosylate leaves a pair of electrons followed by the nucleophilic attack on the reacting molecule to form a new
Lithium aluminum hydride is a strong reducing agent. Alcohols or ethers are the
Oxirane that is commonly called as ethylene oxide is a cyclic ether. Oxirane that is commonly called as ethylene oxide is a cyclic ether.
Answer to Problem 26P
Solution:
a) The principal organic products of the given reaction are as follows:
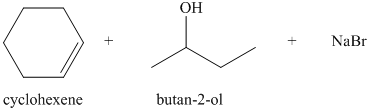
b) The principal organic products of the given reaction are as follows:
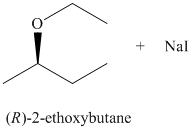
c) The principal organic product of the given reaction is as follows:
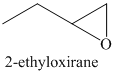
d) The principal organic product of the given reaction is as follows:
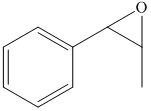
e) The principal organic product of the given reaction is as follows:
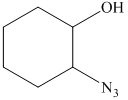
f) The principal organic product of the given reaction is as follows:
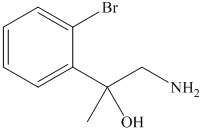
g) The principal organic product of the given reaction is as follows:
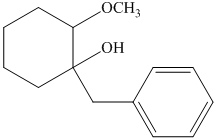
h) The principal organic product of the given reaction is as follows:
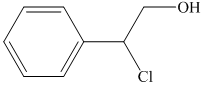
i) The principal organic product of the given reaction is as follows:
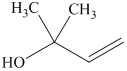
j) The principal organic product of the given reaction is
k) The principal organic product of the given reaction is as follows:
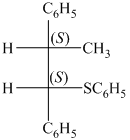
Explanation of Solution
a) Principle organic product formed in the given reaction.
The starting reagents of the given reaction are bromocyclohexane and sodium butan-2-olate. Sodium butan-2-olate is a strong base and bromocyclohexane is a secondary alkyl bromide. Therefore,
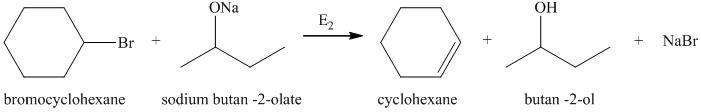
b) Principle organic product formed in the given reaction.
The starting reagents of the given reaction are iodoethane and sodium-(R)-butan-2-olate. They react with each other to form (R)-2-ethoxybutane and sodium iodide. The complete chemical reaction is shown below.
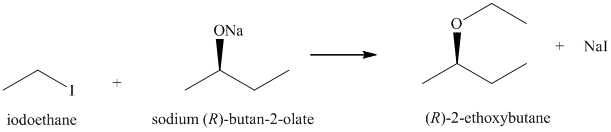
c) Principle organic product formed in the given reaction.
The starting reagent of the given reaction is a halohydrin, 1-bromobutan-2-ol which reacts with a base to form 2-ethyloxirane. The complete chemical reaction is shown below.
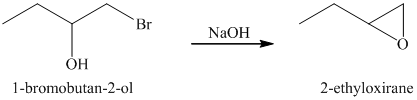
d) Principle organic product formed in the given reaction.
The starting reagent of the given reactions are prop-1-enylbenzene and perbenzoic acid. They react with each other to form 2-methyl-3-phenyloxirane. The complete chemical reaction is shown below.
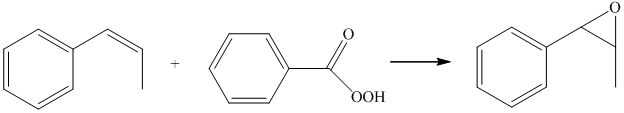
e) Principle organic product formed in the given reaction.
The starting reagent of the given reactions is an
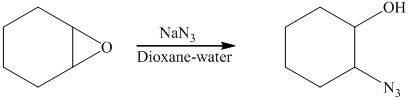
f) Principle organic product formed in the given reaction.
In the presence of the base ammonia, opening of the epoxide ring takes place at less substituted carbon. The complete chemical reaction is shown below.
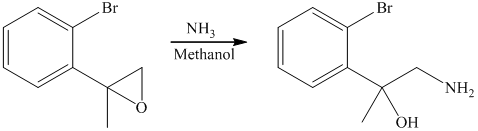
g) Principle organic product formed in the given reaction.
Sodium methoxide is a strong base and in the presence of this strong base and methanol, opening of the epoxide ring takes place at less substituted carbon. The complete chemical reaction is shown below.
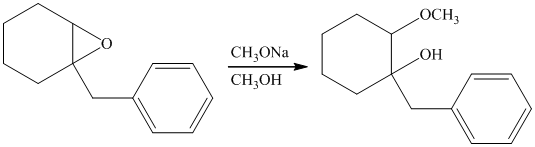
h) Principle organic product formed in the given reaction.
Hydrochloric acid is a strong acid and in the presence of this strong acid and
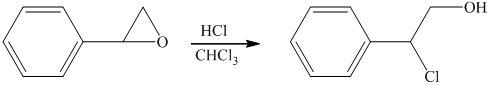
i) Principle organic product formed in the given reaction.
Lithium aluminium hydride is a strong base and in the presence of this strong base and diethyl ether, opening of the epoxide ring takes place at less substituted carbon. The complete chemical reaction is shown below.
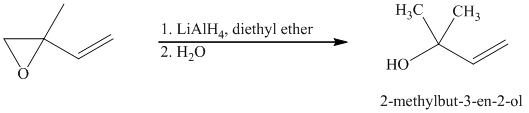
j) Principle organic product formed in the given reaction.
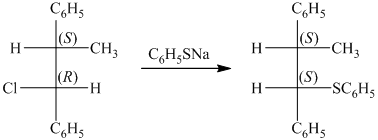
The given starting material is a tosylate. It is a good leaving group. It leaves with a pair of electrons followed by the nucleophilic attack on sodium butane thiolate to form a new
k) Principle organic product formed in the given reaction.
The given starting material is a secondary alkyl halide. It reacts with
Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry - Standalone book
- Give the configuration of each of the following reaction products.arrow_forwardGive the major organic products for each of the following reactions.arrow_forwardPredict the major products of both organic reactions. Be sure to use wedge and dash bonds to show the stereochemistry of the products when it's important, for example to distinguish between two different major products.arrow_forward
- Synthesis. Supply the reagents required to accomplis the following syntheses. Indicate the relative stereochemistry, where applicable, of the products obtained at each step.arrow_forwardPredict the major organic product of each of the following reactions or provide the reagent needed to complete each transformation.arrow_forwardProvide the major organic product for each of the following reactions.arrow_forward
- Predict the major organic product from each of the following reactions.arrow_forwardComplete the following reactions, clearly indicating regio-and stereochemistry where applicable. In cases, where ortho-and para-products are formed, draw both.arrow_forwardGive the major organic product generated by each of the following reactionsarrow_forward
- Predict the major product(s) for each of the following thermal electrocyclic reactions. Justify your answer with MO theory. Heat Heat Heatarrow_forwardGive the major product of the following reactions with the appropriate stereochemistry and Regiochemistry.arrow_forwardCan you please predict the MAJOR product or provide the reagents/conditions for the following reactions? Please indicate racemic if it is and if there is no reaction, please specify that as well. ?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





