
(a)
Interpretation:
The principal organic compound that is expected when
Concept introduction:
The addition reaction of the
Answer to Problem 17.23AP
The principal organic compound that is formed when
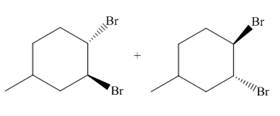
Explanation of Solution
The reaction of
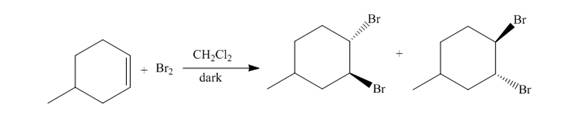
Figure 1
The addition of the bromine molecule on the double bond of the alkene takes place to give the dibromo product. The addition of bromine to the double bond is the anti-addition.
The principal organic compound that is formed when
(b)
Interpretation:
The principal organic compound that is expected when
Concept introduction:
The addition reactions of the alkene are very well known reactions. The electron density on the alkene double bond makes it nucleophilic. The alkene undergoes varieties of addition reaction via different mechanisms.
Answer to Problem 17.23AP
The principal organic compound that is formed when
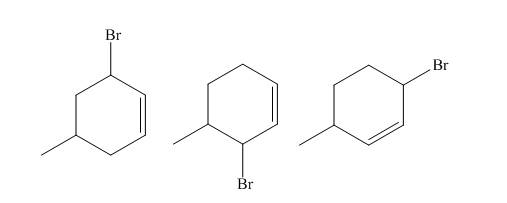
Explanation of Solution
The reaction of
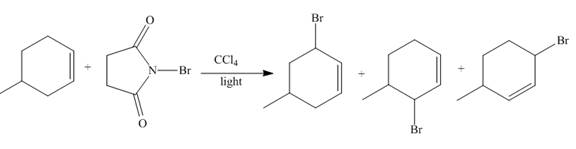
Figure 2
The reaction of an alkene with N-bromosuccinimide in the presence is the free-radical reaction. In this reaction, allylic bromination is observed instead of an addition to the alkene. The N-bromosuccinimide acts as radical initiator. This molecule breaks the
The principal organic compound that is formed when
(c)
Interpretation:
The principal organic compound that is expected when product(s) of part (b) undergo solvolysis in aqueous acetone is to be stated.
Concept introduction:
The nucleophilic substitution reactions are the reactions in which a nucleophile attacks at the electrophilic carbon and eliminates another group. The
The
Answer to Problem 17.23AP
The principal organic compounds that are formed when product(s) of part (b) undergo solvolysis in aqueous acetone are shown below.
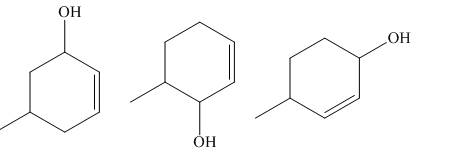
Explanation of Solution
The products of part (b) are shown below.
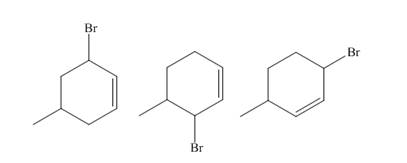
Figure 3
The reaction of product(s) of part (b) on solvolysis in aqueous acetone is shown below.
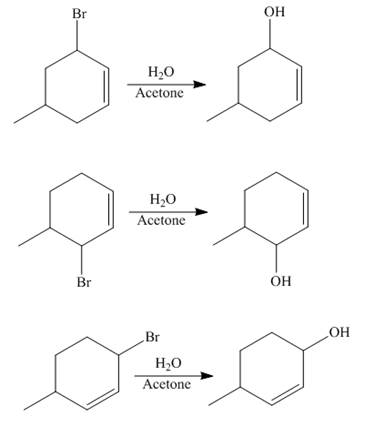
Figure 4
The nucleophilic substitution reaction takes place when solvolysis of products of part (b) is done. The bromine group is substituted by the hydroxyl group giving rise to the formation of an allylic alcohol.
The principal organic compound that is formed when product(s) of part (b) undergo solvolysis in aqueous acetone is shown in Figure 4.
(d)
Interpretation:
The principal organic compound that is expected when product(s) of part (b) are reacted with
Concept introduction:
The reaction of an
Answer to Problem 17.23AP
The principal organic compounds that are formed when product(s) of part (b) are reacted with
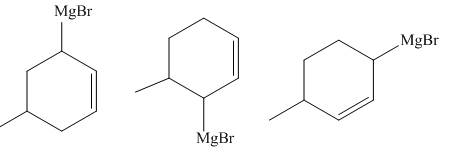
Explanation of Solution
The products of part (b) are shown below.

Figure 3
The reaction that occurs when product(s) of part (b) are reacted with
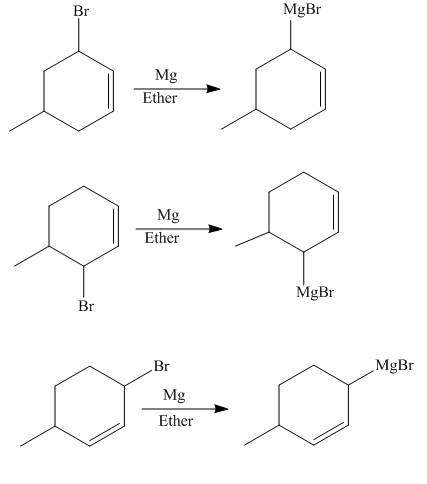
Figure 5
The allylic halide also undergoes the same kind of reactions as the alkyl halide with magnesium metal in dry ether. They also lead to the formation of Grignard reagent but this time with the allylic group.
The principal organic compounds that are formed when product(s) of part (b) are reacted with
(e)
Interpretation:
The principal organic compound that is expected when product(s) of part (d) are reacted with
Concept introduction:
The nucleophilic substitution reactions are the reactions in which a nucleophile attacks at the electrophilic carbon and eliminates another group. The rate of reaction depends upon the nucleophilicity and concentration of the nucleophile.
The
Answer to Problem 17.23AP
The principal organic compounds that are formed when product(s) of part (d) are reacted with
Explanation of Solution
The products of part (d) are shown below.

Figure 6
The reaction of product(s) of part (d) with
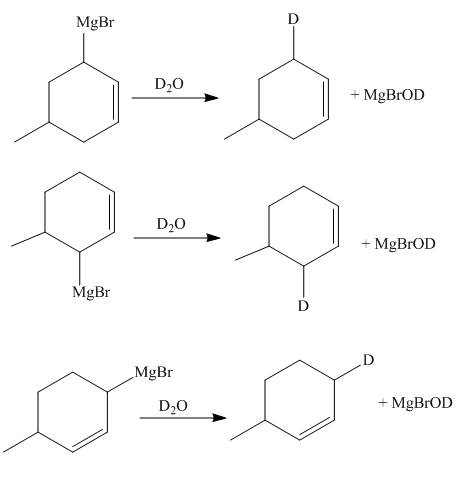
Figure 7
The Grignard reagents are highly reactive towards moisture. The carbon chain on the Grignard reagent gets the hydrogen in place of
The principal organic compounds that are formed when product(s) of part (d) are reacted with
Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry
- Provide the principle organic product for the following reactions. If more than one product is formed indicate which one you expect to be the major product.arrow_forwardBecause bromocyclohexane is a secondary alkyl halide, both cyclohexanol and cyclohexene are formed when the alkyl halide reacts with hydroxide ion. Suggest a method to synthesize cyclohexanol from bromocyclohexane that forms little or no cyclohexene.arrow_forwardWhich of the reaction conditions could afford the following transformation?arrow_forward
- A problem often encountered in the oxidation of primary alcohols to acids is that esters are sometimes produced as by-products. For example, oxidation of ethanol yields acetic acid and ethyl acetate: Propose a mechanism to account for the formation of ethyl acetate. Take into account the reversible reaction between aldehydes and alcohols:arrow_forwardHeterocyclic compounds can undergo nitration reactions with some regioselectivity, giving products in majority. From the following reaction give the raw material from which it is formed and the mechanism under which the transformation takes place.arrow_forwardWhich products are obtained by the following reactions (a) and (b)? Specify the expected main product, if several products can be formed.arrow_forward
- Elimination of HBr from 2-bromobutane affords a mixture of but-1-ene and but-2-ene. With sodium ethoxide as base, but-2-ene constitutes 81% of the alkene products, but with potassium tert-butoxide, but-2-ene constitutes only 67% of the alkene products. Offer an explanation for this difference.arrow_forwardCompound F may be synthesised by the method attached: When 2-chloropropane treated with NaOH and 1-chloropropane treated with NaOH separately produce two different functional groups. Provide both reactions and explain the two different functional groups produced.arrow_forwardAn organic compound A of unknown structure was found to have a molecular formula C8H16. When A was poured in water and heated, compound B having a molecular formula C8H18O was formed. B upon heating with sulfuric acid was converted to C as the major product which is identical to A. Ozonolysis of C gave one molecule each of two different products D and E, both having a molecular formula C4H8O. Write the reactions involved and determine the structure of A,B,C,D and E.arrow_forward
- Predict-the-Product. Draw the structure(s) of the major organic product(s) formed in the following reactions. Please indicate stereochemistry where appropriate.arrow_forwardRank the following hydrocarbons in order of decreasing rates of light-catalyzed bromination. Give the theoretical explanation for the order. Also, give the major bromination product for each.arrow_forwardA) Provide the reagent and reaction mechanism to show how the reactants and products in the following reaction can interconvert B) under what conditions would the reaction I) favour reactants, II) favour the products and III) why?arrow_forward
