
Concept explainers
a)
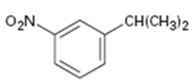
Interpretation:
The
Concept introduction:
The benzene ring is inert to even strong oxidizing agents like KMnO4 and Na2Cr2O7. However the alkyl side chains with benzylic hydrogen atoms are readily oxidized by these reagents into carboxyl groups. Compounds having no benzylic hydrogen atoms in the side chain are not oxidized.
To give:
The aromatic substance obtained as the product, when m-nitroisopropyl benzene is oxidized with KMnO4.
b)
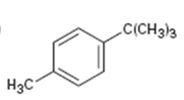
Interpretation:
The aromatic substance obtained as the product, when p-tert-butyltoluene is oxidized with KMnO4, is to be given.
Concept introduction:
The benzene ring is inert to even strong oxidizing agents like KMnO4 and Na2Cr2O7. However the alkyl side chains with benzylic hydrogen atoms are readily oxidized by these reagents into carboxyl groups. Compounds having no benzylic hydrogen atoms in the side chain are not oxidized.
To explain:
The aromatic substance obtained as the product when p-tert-butyl toluene is oxidized with KMnO4.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- What is the function of CH2Cl2 in the bromination reactions? Why can it fulfil this rolearrow_forwardPredict the major products or provide starting materials/reaction reagents for the following reactions.arrow_forward3) Suggest a suitable pathway for the following reactions: 1) H N- N-N Ph Light N N Oarrow_forward
- An electron-rich carbon reacts with an electrophile, symbolized as E+. Explain this ?arrow_forwardGive the expected major product of the following reaction: OH 1) LIAIH,arrow_forwardWhat elimination side reaction can occur from the following reaction in the presence of sulfuric acid? HINT: The product is an alkene but not 1-butene and the reaction has more than one step.arrow_forward
- Four alkenes are formed from the E1 reaction of 3-bromo-2,3-dimethylpentane and methanol. Draw the structures of the alkenes and rank them according to the amount that would be formed.arrow_forwardWrite the reaction and all possible products (E2 and SN2) that could occur for the following:arrow_forwardYou are planning to carry out a reaction between propyne, CH3C≡CH and sodium amide, NaNH2. You also need to choose an appropriate solvent for carrying out the reaction. Would ethanol be suitable for this purpose? Explain your rationale clearly.arrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
