
Concept explainers
(a)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the
functional groups and naming of substituents is done in an alphabetical order.
(a)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
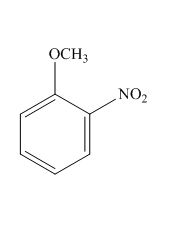
Figure 1
(b)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(b)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
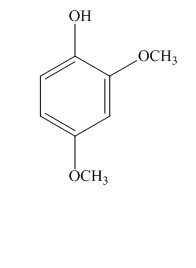
Figure 2
(c)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(c)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
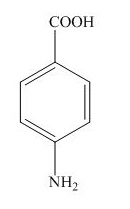
Figure 3
(d)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(d)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
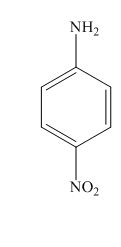
Figure 4
(e)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(e)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
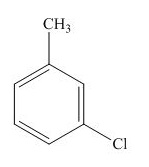
Figure 5
(f)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(f)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
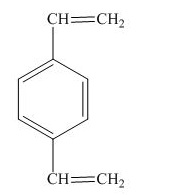
Figure 6
(g)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(g)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
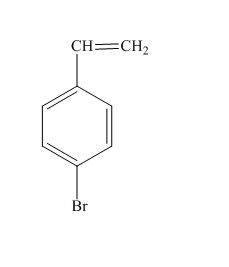
Figure 7
(h)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(h)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
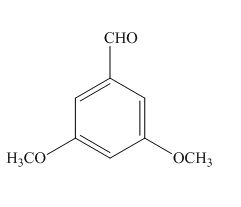
Figure 8
(i)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(i)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
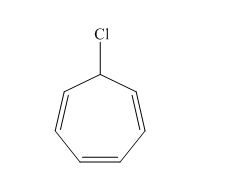
Figure 9
(j)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(j)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
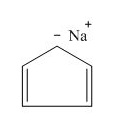
Figure 10
(k)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(k)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
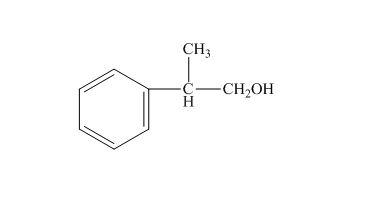
Figure 11
(l)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(l)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
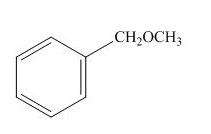
Figure 12
(m)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(m)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
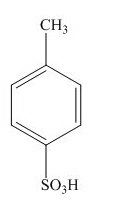
Figure 13
(n)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(n)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
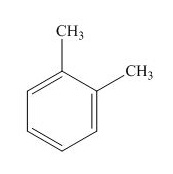
Figure 14
(o)
Interpretation:
The structure of the given compound is to be drawn.
Concept introduction:
Rules for the nomenclature of benzene derivatives are as follows.
- Prefix is added to benzene for the naming of the given compound.
- For the disubstituted compounds numbering is done in such a manner that the substituents occupy the lowest position number.
- Numbering is done on the basis of the priority of the functional groups and naming of substituents is done in an alphabetical order.
(o)
Answer to Problem 16.26SP
The structure of the given compound
Explanation of Solution
The name of the given compound is
The structure for the given compound is shown below.
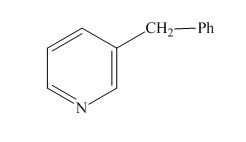
Figure 15
Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
- Give the structure corresponding to each name.a. cyclohexyl propanoateb. cyclohexanecarboxamidec. benzoic propanoic anhydrided. 3-methylhexanoyl chloride e. octyl butanoatef. N,N-dibenzylformamidearrow_forward6. Draw the structure of the following compounds. a. cyclobutanecarboxylic acid b. 3,3-dimethylpentanedioic acid c. 4-aminopentanoic acid d. 2-methylcyclohexanecarboxylic acid e. m-chlorobenzoic acidarrow_forwardPlease explain the chosen letter. Benzoic acid reacts with conc. HNO3 and conc.H2SO, to give a. o-nitrobenzoic acid b. p-nitrobenzoic acid c. m-nitrobenzoic acid d. p-dinitrobenzoic acidarrow_forward
- Cyclopentanone is treated with chlorine (Cl2) in the presence of acid (H*). What product is formed? A. Chlorocyclopentane B. 1-chloro-1-hydroxycyclopentane C. 2-chlorocyclopentanone D. 3-chlorocyclopentanonearrow_forwardB. Draw structures corresponding to the following names. 1. 3-methylbutanal 7. 4-chloro-2-pentanone 2. o-hydroxybenzoic acid 8. phenylacetaldehyde 3. 3-chloro-4-cyanopentanoic acid 9. cis-3-tert-butylcyclohexanecarbaldehyde 4. 3-methyl-3-butenal 10. 2-(1-chloroethyl)-5-methylheptanal 5. 2,3-dimethylhexanoic acid 11. trans-1,2-cyclobutanedicarboxylic acid 6. 2-hexen-4-ynoic acid 12. heptanedioic acidarrow_forwardCis-but-2-ene has a different set of properties compared to trans-but-2-ene. the melting points of these two compounds is influence primarily by what factor? a. Dipole moment b. Hydrogen Bonding c. Symmetry of molecule d. Van de Waals interaction Which electrophilic aromatic substitution reaction is described when aniline is transformed into para-aminotolouene? a. Halogenation b. Nitration c. Oxidation d. Friedel Crafts alkylation The following hydrocarbons will exhibit electrophilic reactions (addition or substitution) involving halogens except: a. ethyne b. ethene c. ethane d. benzenearrow_forward
- 1. Which carbonyl group of the given compound is most reactive for nucleophilic addition reaction? a. All have equal reactivity b. Carbonyl Group 1 c. Carbonyl Group 2 d. Carbonyl Group 2. An aldehyde commonly exhibits a nucleophilic addition type of reaction. When a nucleophile attacks a carbonyl carbon, what happens to the oxygen atom in the structure? Refer to the structure below. ~ a. Oxygen atom becomes more electronegative. b. Oxygen atom obtains a net negative charge. c. Oxygen atom transforms to an alkoxide group. d. Oxygen atom acts as the new electrophile. 3. Assign the trivial name of the structure below. a. Diphenylketone b. Benzyl phenylketone c. Diphenyl aldehyde d. Benzyl phenyl aldehydearrow_forwardReagents a. C6H5CHO b. NaOH, ethanol h. BrCH2CH=CH2 i. Na* OEt, ethanol j. Br2, H* k. K* t-BuO c. Pyrrolidine, cat. H* d. H2C=CHCN e. H3O* f. I. CH2(CO2ET)2 -CH2CH2CN LDA m. heat g. ELOC(=0)CO2ET Select reagents from the table to synthesize this compound from cyclopentanone. Enter the letters of the chosen reagents, in the order that you wish to use them, without spaces or punctuation (i.e. geda).arrow_forwardCH₂ CH₂ H KMnO4 H₂C 61. What type of reaction is catalyzed based on the chemical equation? A. Acid-catatalyzed dehydration B. Oxidation C. Reduction D. Williamson synthesis 62. What is the expected final product of this reaction? A. 3-methylbutanoic acid B. 2-methylbut-3-enoic acid C. 3-methyl-2-oxobutanoic acid D. 2-methylbutanoic acid 63. What is the functional group of the intermediate product in the reaction? A. Ketone B. Carboxylic acid C. Aldehyde D. Ether 个 5 ▸arrow_forward
- 1. An alkene reacts with water with an acid catalyst results into a formation of: A. Aldehyde B. Ketone C. Alcohol D. Ester 2. 3-Methylhexanal with K2Cr2O7 will yield: A. 3-Methyl-1-hexanol B. 3-Methylhexanoic acid C. 3-Methyl-1-hexanone D. 3-Methyl-1-hexanethiol 3. This is a reverse process of Hydration reaction: A. Oxidation reaction B. Reduction reaction C. Dehydration reaction D. Hydration reaction 4. Acetic acid reacts with a strong base forms: A. Salt B. Water C. Salt and Water D. No reaction 5. Ketones can be further oxidized with benedict's solution into: A. Alcohol B. Aldehyde C. Catalysts D. No reactionarrow_forwardShow how to convert cyclohexanol to these compounds. a. Cyclohexene b. Cyclohexane c. Cyclohexanone d. Bromocyclohexanearrow_forwardWhich of the following reacts fastest in nucleophilic addition reaction? a. Acetic acid b. Ethanol c. Formaldehyde d. Benzaldehyde e. Acetone How can a water insoluble acid be converted into a water soluble compound? a. By reacting it with an alcohol. b. By reacting it with hydrochloric acid. c. By reacting it with nitric acid. d. None of the mentioned method. e. By reacting it with sodium bicarbonatearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





