
(a)
Interpretation:
The CN stretching mode that absorbs the higher-frequency IR photons is to be indicated for given pair of compounds. The reason for it is to be explained.
Concept introduction:
We simplify the picture of molecular vibrations by considering the ball-and-spring model, which treats bonds as simple springs that connect atoms together. According to Hooke’s law, the spring vibrates at a particular frequency (
Answer to Problem 15.19P
The CN stretching mode that absorbs the higher-frequency IR photons due to strong and stiffer bond is indicated below,
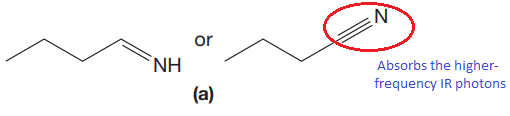
Explanation of Solution
The given pair of compounds is,
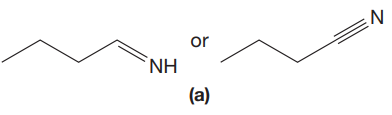
The triple bonds tend to be stronger and stiffer than double bonds. A stronger and stiffer bond tends to lead to a higher vibrational frequency. In given pair of compounds, with the faster vibration, the
The CN stretching mode that absorbs the higher-frequency IR photons is shown below,

The CN stretching mode that absorbs the higher-frequency IR photons is indicated on the basis of the relationship between strength and stiffness of the bond and vibrational frequency.
(b)
Interpretation:
The CN stretching mode that absorbs the higher-frequency IR photons is to be indicated for given pair of compounds. The reason for it is to be explained.
Concept introduction:
We simplify the picture of molecular vibrations by considering the ball-and-spring model, which treats bonds as simple springs that connect atoms together. According to Hooke’s law, the spring vibrates at a particular frequency (
Answer to Problem 15.19P
The CN stretching mode that absorbs the higher-frequency IR photons due to strong and stiffer bond is indicated below,
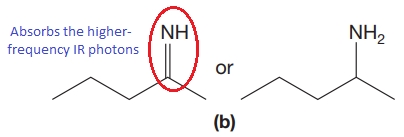
Explanation of Solution
The given pair of compounds is,
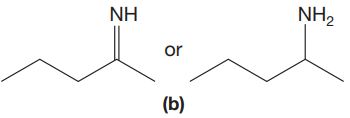
The double bonds tend to be stronger and stiffer than single bonds. A stronger and stiffer bond tends to lead to a higher vibrational frequency. In given pair of compounds, with the faster vibration, the
The CN stretching mode that absorbs the higher-frequency IR photons is shown below,

The CN stretching mode that absorbs the higher-frequency IR photons is indicated on the basis of the relationship between strength and stiffness of the bond and vibrational frequency.
(c)
Interpretation:
The CN stretching mode that absorbs the higher-frequency IR photons is to be indicated for given pair of compounds. The reason for it is to be explained.
Concept introduction:
We simplify the picture of molecular vibrations by considering the ball-and-spring model, which treats bonds as simple springs that connect atoms together. According to Hooke’s law, the spring vibrates at a particular frequency (
Answer to Problem 15.19P
The CN stretching mode that absorbs the higher-frequency IR photons due to lower mass is indicated below,
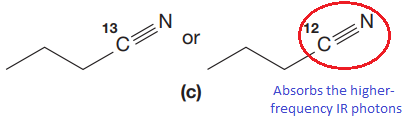
Explanation of Solution
The given pair of compounds is,
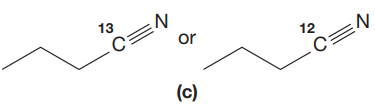
In both compounds there is
But
The CN stretching mode that absorbs the higher-frequency IR photons is shown below,

The vibrational mode that absorbs at a higher frequency in the IR region is determined on the basis of the relationship between mass and vibrational frequency.
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Problem (a) Estimate the energy of the electrons that we need to use in an electron microscope to resolve a separation of 0.27 mm. (b) In a scattering of 2 eV protons from a crystal, the fifth maximum of the intensity is observed at an angle of 30°. Estimate the crystal's planar separation.arrow_forwardBased on your answer to below Problem, do you thinkthe compound shown here should have a significantdipole moment? If so, in which direction does it point? The molecule shown here has quite a large dipole, asindicated in its electrostatic potential map. Explain why.Hint: Consider various resonance structures.arrow_forwardLet us discuss whether H-H bond should have IR activity in spectrum. If, what is the approximate wavelength that peak locates, if not, explain why.arrow_forward
- (Answer with G explanation please give)arrow_forwardProblem. Calculate the dipole moment of Glycine molecule using partial charge and positions of atoms. In this problem, How can I get the partial charge of the atoms? Should I find the reference or appendix?arrow_forwardGive both answer otherwise don't asaparrow_forward
- 2. According to the Beer-Lambert law absorbance is proportional to concentration at each wavelength. In principle, one could choose any wavelength for quantitative estimations of concentration. However, we generally select the wavelength of maximum absorbance for a given sample and use it in our absorbance measurements. Especially, one should avoid the steep portions of the spectrum. Why? 3. Which concentration range a dilute or a concentrated one - is chosen for accurate determinations of unknown concentrations?arrow_forwardPlease answer with explanationsarrow_forwardWhy is there no coupling between the a and c protons or between the b and c protons in the cis and trans alkenes shown in Figurearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

