
Concept explainers
(a)
Interpretation:
The bond length of the indicated
Concept introduction:
Weak resonance contributors give some single-bond character to the two terminal
Answer to Problem 14.33P
In
Explanation of Solution
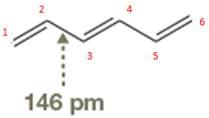
The bond length of
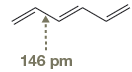
The normal
The amount of shortening of
(b)
Interpretation:
The bond length of the indicated
Concept introduction:
Resonance contributors give some double-bond character to the
Answer to Problem 14.33P
The occupied
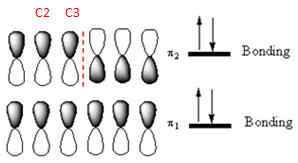
Explanation of Solution
The bond length of
The occupied
Out of these, the occupied

The occupied
(c)
Interpretation:
The bond length of the indicated
Concept introduction:
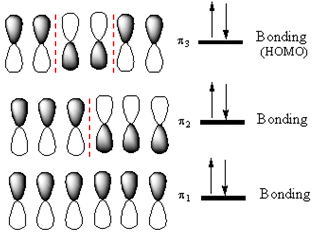
The highest energy MO of
Answer to Problem 14.33P
If an electron were promoted from the HOMO to the LUMO, the length of the
Explanation of Solution
The
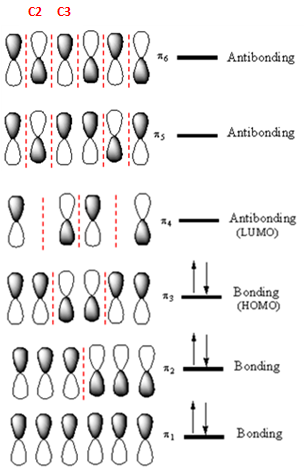
If an electron is promoted from HOMO (
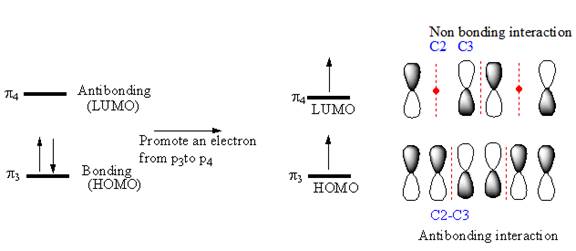
Therefore, the removal of an electron from the HOMO contributes to a shorter C2-C3 bond.
If an electron were promoted from the HOMO to the LUMO, the length of the
Want to see more full solutions like this?
Chapter 14 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- 2- 7. A Lewis electron-dot diagram of the oxalate ion, C₂0₂², is shown. (a) Identify the hybridization of the valence orbitals of either carbon atom in the oxalate ion. (b) Silver oxalate, Ag₂C₂O4(s), is slightly soluble in water. The value of Köp for Ag₂C₂04 is 5,40 × 10-¹². (i) Write the expression for the solubility-product constant, Kp, for Ag₂C₂04. (ii) Calculate the molar solubility of Ag₂C₂O4 in neutral distilled water.arrow_forwardDraw the following Lewis structures using condensed formulas. (a) нннн | | | | Н—С—С—С—С—н (b) нннн (c) | | | | Н-С—С—С—С—н H-C-H H H H H H H нн H H-C-C=ċ-ċ-H H. (d) H. (е) H (f) нн H. H-C-C Н-с—о—С-с-н H- C-C H-C. нн H. H-C. C- H H H. H нн H I- HIC-Iarrow_forwardThe pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.(b) How many MOs are there in the molecular orbital picture of the pentadienyl radical?arrow_forward
- Draw the shapes of the following molecules and ions in 3-dimension. Show clearly any lone pairs of electrons on the central atom, state the number of bond pairs and lone pairs of electrons on the central atom and name the shape of the molecule or ion. (a) CH3+, methyl carbocation (b) HOBr, bromic (I) acid (c) NCl3, nitrogen trichloride Provide everything stated in the instructions for each compound.arrow_forwardFor each of the following structures, determine the hybridization requested and whether the electrons will be delocalized: (a) Hybridization of each carbon н :0: Н | || | Н—с—с——н H. (b) Hybridization of sulfur (c) All atoms EN: H.arrow_forwardAverage Bond Dissociation Energies at 298 K Bond Energy, kJ mol1 H-H 432 O=0 495 H-O 467 The tables above contain information for determining thermodynamic properties of the reaction below. 2H,(g) + 02(8) - 2H,O(g) (a) Calculate the AH° for the reaction above, using the table of average bond dissociation energy. (b) Use the table below to calculate the entropy for the formation of water. 2H, + 02 - 2H,O Substance S°(J/mol· K) H2 131.0 H20 69.91 O2 205.0 (c) Calculate the value of AG°298K for the synthesis of water.arrow_forward
- 2. The full structural formulae of three organic compounds, P, Q and R, are shown below. H H H H HH HHHH Н-С -с- Н H-C- C - C = C – H H - C - C = C – C – H H H H H Q P (a) State one similarity between P, Q and R in terms of their molecular formulae. (b) Name the homologous series that compounds P, Q and R belong to. (c) State one similarity between Q and R in terms of chemical bonding id) Which of these compounds are isomers? Explain your answer.arrow_forwardThiocyanic acid (HSCN) is an inorganic acid (pKa = 1.1 at 25 °C) that can be classified as “strong” largely due to resonance stabilization of its conjugate base, thiocyanate (SCN– ). In the space provided below, draw Lewis electron dot structures of SCN- and all of its important resonance forms.arrow_forwardQ. 9a) Provide additional resonance structures for the following species. Remember to show lone pairs & formal charges where applicable + [ :N=N-F: +→ [:0-C!-O: → Q. 9b) Draw the Lewis Structure for NCCH2CO2NHCH3, determine the number of sigma bonds & the number of pi bonds present, and indicate the bond angle value around each carbon atom, each nitrogen atom, and each oxygen atom.arrow_forward
- The pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.(b) How many MOs are there in the molecular orbital picture of the pentadienyl radical?(c) How many nodes are there in the lowest-energy MO of the pentadienyl system? How many in the highest-energy MO?(d) Draw the MOs of the pentadienyl system in order of increasing energyarrow_forwardThe pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.(b) How many MOs are there in the molecular orbital picture of the pentadienyl radical?(c) How many nodes are there in the lowest-energy MO of the pentadienyl system? How many in the highest-energy MO?(d) Draw the MOs of the pentadienyl system in order of increasing energy. (continued)762 CHAPTER 15 Conjugated Systems, Orbital Symmetry, and Ultraviolet Spectroscopy(e) Show how many electrons are in each MO for the pentadienyl radical (ground state).(f) Show how your molecular orbital picture agrees with the resonance picture showing delocalization of the unpairedelectron onto three carbon atoms.(g) Remove the highest-energy electron from the pentadienyl radical to give the pentadienyl cation. Which carbon atomsshare the positive charge? Does this picture agree with the resonance picture?(h) Add an…arrow_forwardUsing a table of average bond enthalpies. Table 6.2 ( Sec. 6-6b), estimate the enthalpy change for the industrial synthesis of methanol by the catalyzed reaction of carbon monoxide with hydrogen.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning



