
Concept explainers
Draw the enol tautomers for each of the following compounds. If the compound has more than one enol tautomer, indicate which one is more stable.
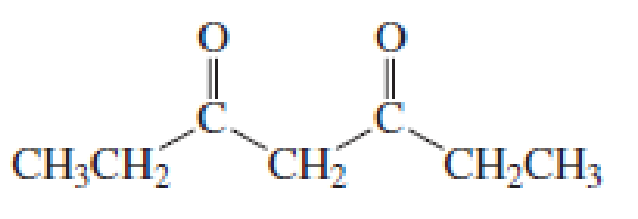
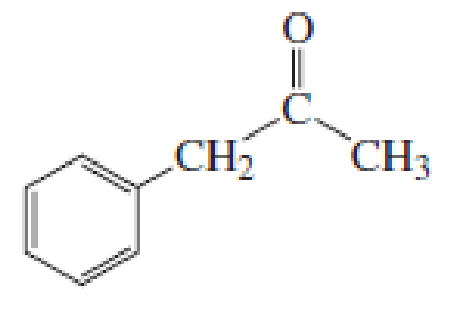
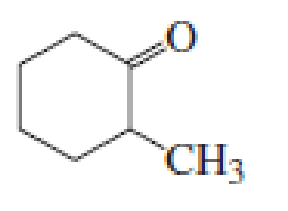
(a)
Interpretation:
The enol tautomer of the given compound has to be drawn and more stable structure has to be identified.
Concept Introduction:
Tautomerism is the ability of a molecule to exist in more than one chemical form.
Tautomers are formed by the migration of a hydrogen atom, accompanied by the switching of a single and neighboring double bond.
The only difference in keto-enol tautomers is the location of hydrogen and double bond.
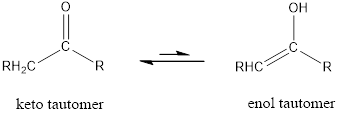
Enol tautomer is much less stable than the keto tautomer.
Enol tautomer is more stable when enol tautomer is aromatic or when the double bonds are conjugated.
Resonance is an electron displacement effect for stabilizing a molecule through delocalization of bonding electrons in the pi orbital.
Delocalized electrons stabilize a compound. The extra stability gains from having delocalized electrons are called resonance stabilization or resonance energy.
Explanation of Solution
Given keto tautomer is,
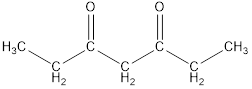
The only difference in keto-enol tautomer is the location of hydrogen and double bond.
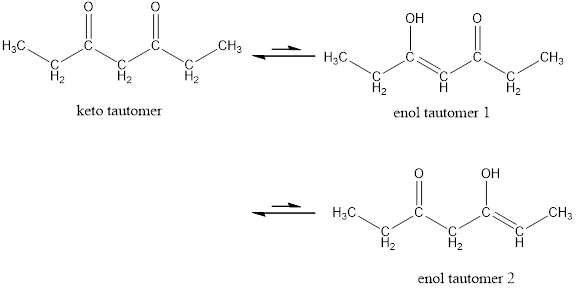
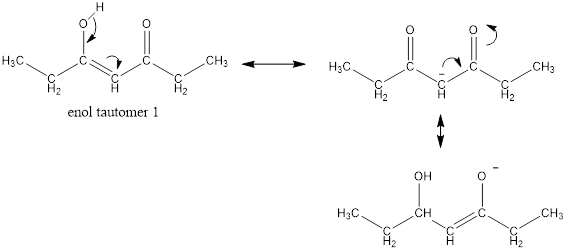
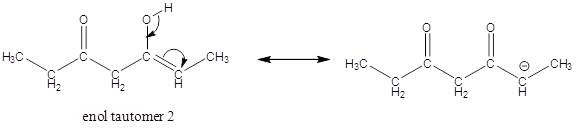
Enol tautomer 1 is more stable than enol tautomer 2.
Enol tautomer 1 can undergo delocalization and are more stable.
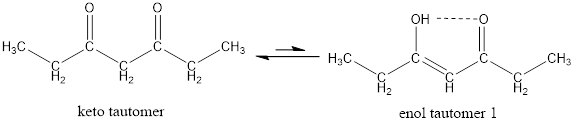
Thus enol tautomer 1 is more stable since it has more resonance structures and also possess intramolecular hydrogen bonding.
(b)
Interpretation:
The enol tautomer of the given compound has to be drawn and more stable structure has to be identified.
Concept Introduction:
Tautomerism is the ability of a molecule to exist in more than one chemical form.
Tautomers are formed by the migration of a hydrogen atom, accompanied by the switching of a single and neighboring double bond.
The only difference in keto-enol tautomers is the location of hydrogen and double bond.

Enol tautomer is much less stable than the keto tautomer.
Enol tautomer is more stable when enol tautomer is aromatic or when the double bonds are conjugated.
Resonance is an electron displacement effect for stabilizing a molecule through delocalization of bonding electrons in the pi orbital.
Delocalized electrons stabilize a compound. The extra stability gains from having delocalized electrons are called resonance stabilization or resonance energy.
Explanation of Solution
Given keto tautomer is,
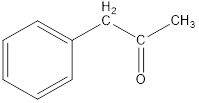
The only difference in keto-enol tautomers is the location of hydrogen and double bond.
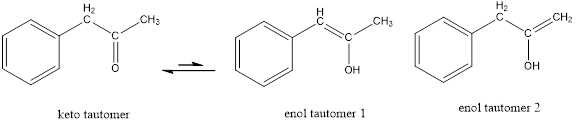
These tautomers undergo resonance and are shown below,
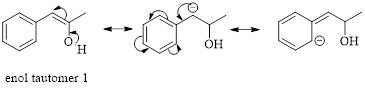
Enol tautomer 1 can undergo delocalization. Enol tautomer 1 is more stable than enol tautomer 2.
The enol tautomer 1 is more stable because there is a conjugation between the double bond and benzene ring. No such conjugation is possible in the enol tautomer 2.
(c)
Interpretation:
The enol tautomer of the given compound has to be drawn and more stable structure has to be identified.
Concept Introduction:
Tautomerism is the ability of a molecule to exist in more than one chemical form.
Tautomers are formed by the migration of a hydrogen atom, accompanied by the switching of a single and neighboring double bond.
The only difference in keto-enol tautomers is the location of hydrogen and double bond.

Enol tautomer is much less stable than the keto tautomer.
Enol tautomer is more stable when enol tautomer is aromatic or when the double bonds are conjugated.
Explanation of Solution
Given keto tautomer is,
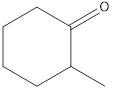
The only difference in keto-enol tautomers is the location of hydrogen and double bond.
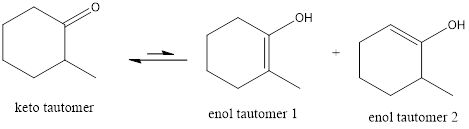

The alkene double bond formed by tautomer 1 is tetra-substituted which is stable than tautomer 2.
Hence, enol tautomer 1 is more stable.
Want to see more full solutions like this?
Chapter 13 Solutions
Essential Organic Chemistry (3rd Edition)
- Draw the product when each compound is treated with either (CH3)2CuLi, followed by H2O, or HC≡CLi, followed by H2O.arrow_forwardOf the following structures, which is the most stable enol form?arrow_forwardDraw the tautomer of this enol. Include all lone pairs. Ignore inorganic byproducts. H3O+ Drawing OHarrow_forward
- Answer the following questions about curcumin, a yellow pigment isolated from turmeric, a tropical perennial in the ginger family and a principal ingredient in curry powder.a.In Chapter 11, we learned that most enols, compounds that contain a hydroxy group bonded to a C=C, are unstable and tautomerize to carbonyl groups. Draw the keto form of the enol of curcumin, and explain why the enol is more stable than many other enols. b.Explain why the enol O—H proton is more acidic than an alcohol O—H proton. c. Why is curcumin colored? d.Explain why curcumin is an antioxidant.arrow_forwardThe major product of the given reaction has the molecular formula C10H16O3. Draw its structure in the most stable tautomeric form.arrow_forwardDraw two enol forms of the following diketonearrow_forward
- Draw the products formed when each compound is treated with HNO3 and H2SO4.State whether the reaction occurs faster or slower than a similar reaction with benzene.arrow_forwardIdentify the best reagents to complete the following reaction. HO, CIarrow_forwardIdentify the correct reagent for each of the following reactions.arrow_forward
- Choose the most correct set of reagents for the following reaction.arrow_forwardDraw the tautomer of this ketone. Include all lone pairs. Ignore inorganic byproducts. :0: H3O+ 0 ✔arrow_forwardDrawing an Enol and a Ketone Formed by Hydration of an Alkyne Draw the enol intermediate and the ketone product formed in the following reaction.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
