
Concept explainers
(a)
Interpretation:
Whether the change
Concept Introduction:
Electron transport chain is a sequence of biochemical reactions in which electrons and hydrogen atoms from the citric acid cycle are transferred to various intermediate carriers and finally reacts with molecular oxygen to form a water molecule.
There are four complexes associated with the electron transport chain that is present in the inner mitochondrial membrane. The four complexes that help in the electron transfer in the electron transport chain are:
Complex I:
Complex II:
Complex III:
Complex IV:
An overview of the electron transport chain is as follows:
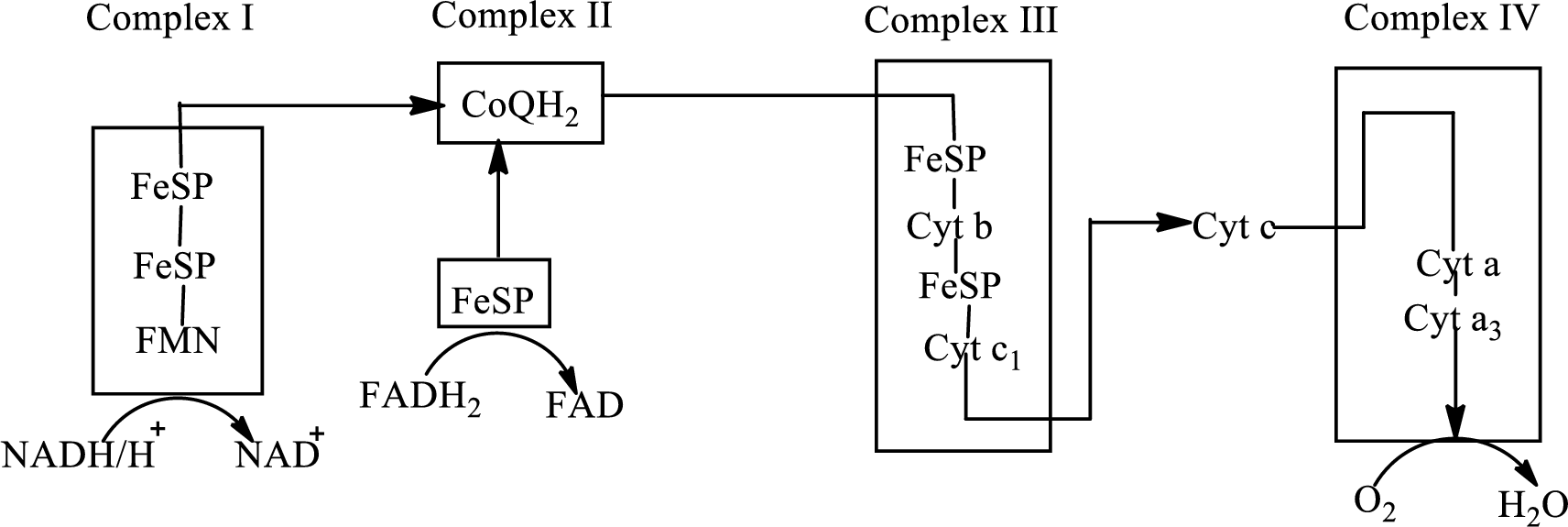
Redox
Here A is oxidized form and AH is reduced form.
(a)
Answer to Problem 12.89EP
The change
Explanation of Solution
(b)
Interpretation:
Whether the change
Concept Introduction:
Electron transport chain is a sequence of biochemical reactions in which electrons and hydrogen atoms from the citric acid cycle are transferred to various intermediate carriers and finally reacts with molecular oxygen to form a water molecule.
There are four complexes associated with the electron transport chain that is present in the inner mitochondrial membrane. The four complexes that help in the electron transfer in the electron transport chain are:
Complex I:
Complex II:
Complex III:
Complex IV:
An overview of the electron transport chain is as follows:
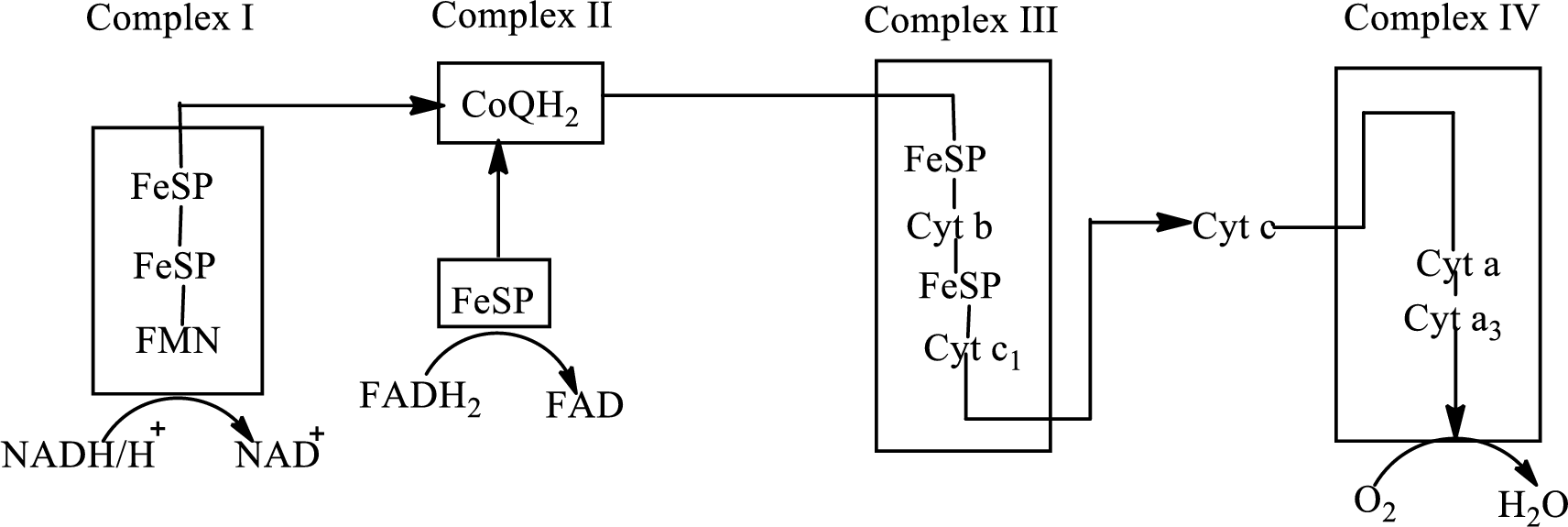
Redox reactions involve oxidation and reduction reaction occurring simultaneously so that one species is oxidized and the other one is reduced. The species that gain hydrogen or electron is known as reduced form and the species that loss hydrogen or electron is known as oxidized form. The general representation of the redox reaction is,
Here A is oxidized form and AH is reduced form.
(b)
Answer to Problem 12.89EP
The change
Explanation of Solution
In complex I, electrons are transferred from the
The change of
(c)
Interpretation:
Whether the change
Concept Introduction:
Electron transport chain is a sequence of biochemical reactions in which electrons and hydrogen atoms from the citric acid cycle are transferred to various intermediate carriers and finally reacts with molecular oxygen to form a water molecule.
There are four complexes associated with the electron transport chain that is present in the inner mitochondrial membrane. The four complexes that help in the electron transfer in the electron transport chain are:
Complex I:
Complex II:
Complex III:
Complex IV:
An overview of the electron transport chain is as follows:
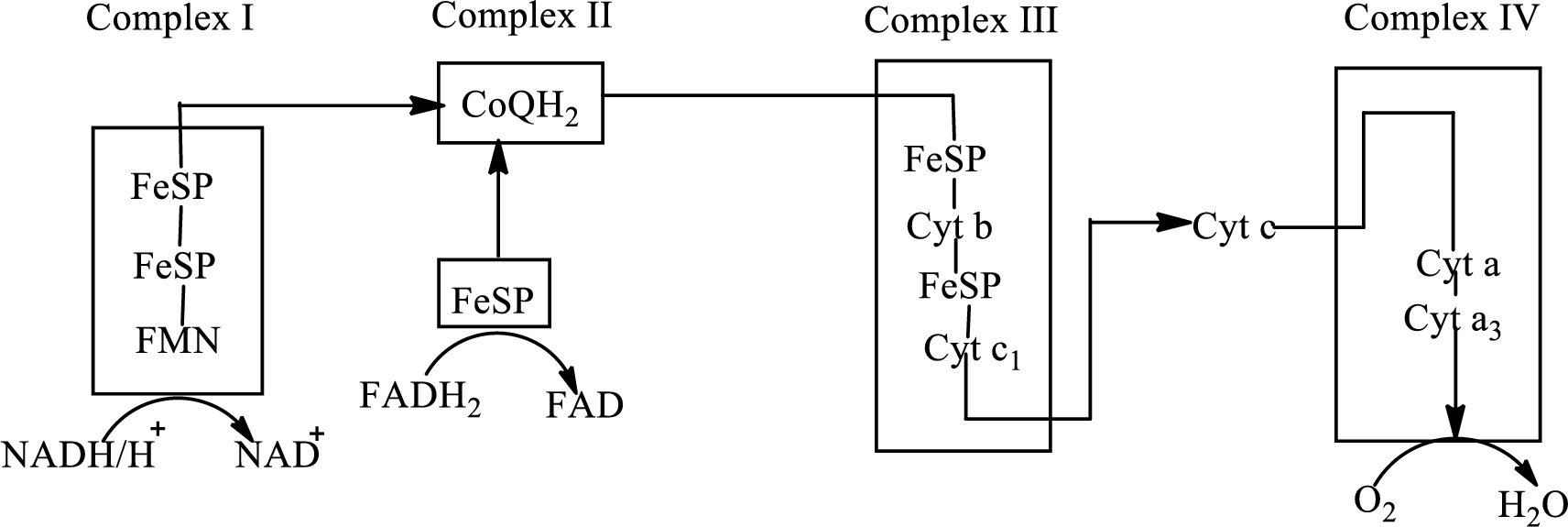
Redox reactions involve oxidation and reduction reaction occurring simultaneously so that one species is oxidized and the other one is reduced. The species that gain hydrogen or electron is known as reduced form and the species that loss hydrogen or electron is known as oxidized form. The general representation of the redox reaction is,
Here A is oxidized form and AH is reduced form.
(c)
Answer to Problem 12.89EP
The change
Explanation of Solution
(d)
Interpretation:
Whether the change
Concept Introduction:
Electron transport chain is a sequence of biochemical reactions in which electrons and hydrogen atoms from the citric acid cycle are transferred to various intermediate carriers and finally reacts with molecular oxygen to form a water molecule.
There are four complexes associated with the electron transport chain that is present in the inner mitochondrial membrane. The four complexes that help in the electron transfer in the electron transport chain are:
Complex I:
Complex II:
Complex III:
Complex IV:
An overview of the electron transport chain is as follows:
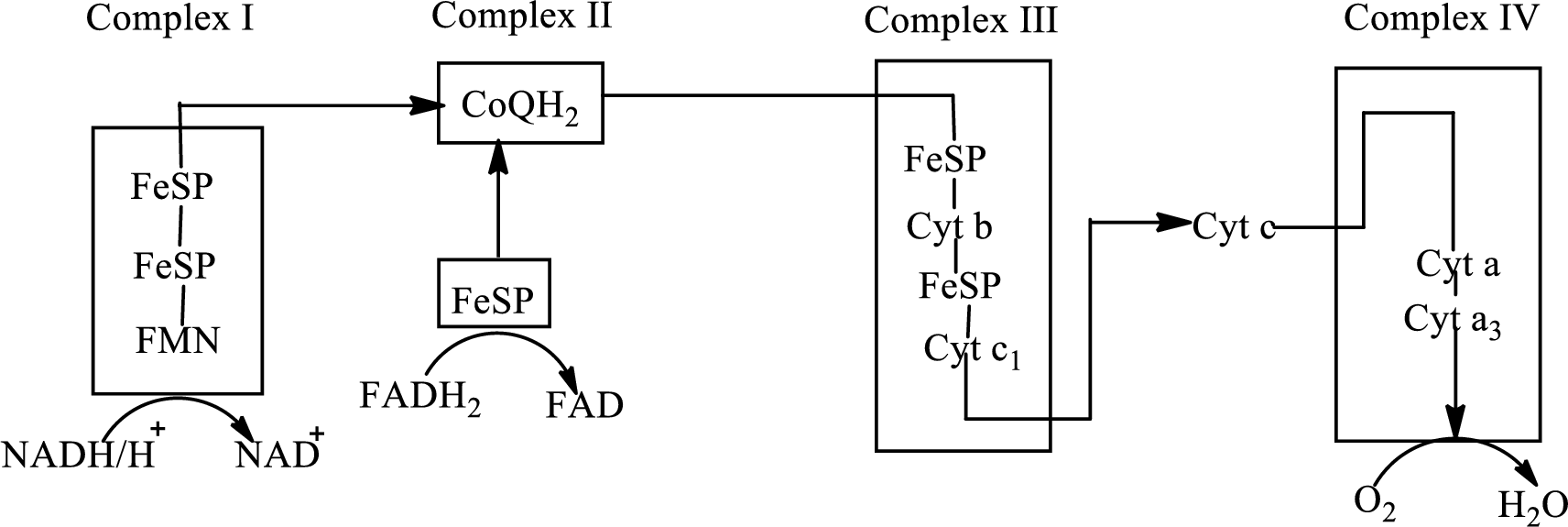
Redox reactions involve oxidation and reduction reaction occurring simultaneously so that one species is oxidized and the other one is reduced. The species that gain hydrogen or electron is known as reduced form and the species that loss hydrogen or electron is known as oxidized form. The general representation of the redox reaction is,
Here A is oxidized form and AH is reduced form.
(d)
Answer to Problem 12.89EP
The change
Explanation of Solution
Want to see more full solutions like this?
Chapter 12 Solutions
Organic And Biological Chemistry
- Indicate whether each of the following substances undergoes an oxidation number increase or decrease in a redox reaction. a. The oxidizing agent b. The reducing agent c. The substance undergoing oxidation d. The substance undergoing reductionarrow_forwardIs the following reaction an oxidation, reduction, or neither? но OH O reduction oxidation neitherarrow_forwardWhich of the following is the best oxidizing agent? NO3 NO2 fumarate succinatearrow_forward
- 26. Calculate AG for the following reaction at 150 °C (attach your solution) C2H4(g) + 3 O2(g) 2 CO2(g) + 2 H2O(g) O 313 kcal O -313 kcal O 312 kcal -312 kcalarrow_forwardHow many electrons are transferred in the given reaction? Ni + 2 HCl → Ni Cl2 + H2arrow_forwardDetermine the oxidation number of each element in these molecule. Put the negative sign in front of any negative oxidation number; do NOT put a (+) in front of any positive oxidation number. I P406 P HNO3 H KCIO₂ K O Z D O Oarrow_forward
- Identify the substances with the corresponding role in the following equation.Ag(s)+H2S→Ag2S(g)+H2(g) A. oxidized B. oxidizing agent C. reduced 1. Ag - a,b, or c? 2. H2S - a,b, or c? 3. H- a,b, or c?arrow_forwardI2O5 (s) + 5 CO (g) -> I2 + 5 CO2 (g) How many electrons are transferred in this reaction?arrow_forwardIn terms of cellular respiration,can anyone tell me what is being oxidized and what is reduced for the below formulas? Let’s take a look at some examples of full redox reactions CO2 (g) + H2 (g) → CO (g) + H2O (g) CH4 (g) + O2 (g) → CO2 (g) + H2O (g) Zn + CuCl2 → ZnCl2 + Zn Na + Cl → NaClarrow_forward
- What is the oxidation number of C in K₂C₂O4? If the oxidation number is positive, just write the number. For example, write "1" for an oxidation number of positive one. If the oxidation number is negative, write the negative sign before the number. For example, write "-1" for an oxidation number of negative one.arrow_forwardWhat will happen if the system is cooled? What will happen if more CO2 (g) is added?arrow_forwardFor the reaction: 2HBr → H2 + Br2 ΔH = -17.4 kcal/mol. And Keq = 32 X 10 20 Is the reaction exothermic or endothermic? Are products or reactants favored? Using Le Chatelier’s Principle, what happens to the equilibrium if the concentration of Br2 is increased? Using Le Chatelier’s Principle, what happens to the equilibrium if the concentration of H2 is decreased?arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

