
Concept explainers
(a)
Interpretation:
The compound
Concept Introduction:
Organic compound is that compound in which carbon is covalently bonded to hydrogen atom or to oxygen or nitrogen atom. For example:
Inorganic compound is the one in which no carbon hydrogen
Answer to Problem 21P
Explanation of Solution
The structure of
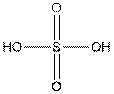
In the structure of
(b)
Interpretation:
The compound
Concept Introduction:
Organic compound is that compound in which carbon is covalently bonded to hydrogen atom or to oxygen or nitrogen atom. For example:
Inorganic compound is the one in which no carbon hydrogen
Answer to Problem 21P
Explanation of Solution
The structure of

In compound
(c)
Interpretation:
The compound
Concept Introduction:
Organic compound is that compound in which carbon is covalently bonded to hydrogen atom or to oxygen or nitrogen atom. For example:
Inorganic compound is the one in which no carbon hydrogen
Answer to Problem 21P
Explanation of Solution
The structure of
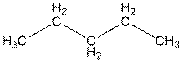
In
Want to see more full solutions like this?
Chapter 11 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Which chemical formulas represent organic compounds and which represent inorganic compounds: (a) H 2SO 4; (b) Br 2; (c) C 5H 12?arrow_forwardClassify compound as organic or inorganic. C4H6O6arrow_forwardName these organic compounds: compound F | CH₂ | I-CH₂-CH₂2-C-C- CH3 Cl F CH₂ CH,— CH,— CH,— CH,— I CI | C1 CH₂-C- CH₂-Cl nam 0 0 0arrow_forward
- Name the following organic compounds: name compound CH3 · CH3 CH - CH; - CH3 · CH,- CH, C - CH,– CH, - 3-ethyl-3-methylpentNe CH2 CH3 CH3 CH,– CH, CH,– CH – CH– CH, CH3arrow_forward2.) Please name B(OH)2arrow_forwardDistinguish between High Density Polyethylene (HDPE), Low Density Polyethylene (LDPE) and Linear Low Density Polyethylene (LLDPE) by giving ONE (1) example each.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





