
Concept explainers
a)
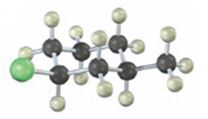
Interpretation:
The IUPAC name of the
Concept introduction:
The longest continuous carbon chain in the molecule is chosen. The chain is numbered from the end which gives lowest number to the substituent either halo or alkyl group present. If different halogens are present they are numbered and listed in the alphabetical order while writing the name. If same two alternatives exist for different substituents, then the chain is numbered from the end that gives lowest number to the substituent that has alphabetical preference.
To give:
The IUPAC name of the alkyl halide shown.
Answer to Problem 14VC
The alkyl halide given is
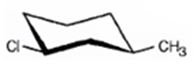
Its IUPAC name is cis- 1- chloro-3-methylcyclohexane.
Explanation of Solution
The compound has a cyclohexane ring with a Cl atom on C1 and a methyl on C3 both at equatorial positions. Hence its name is cis- 1- chloro-3-methylcyclohexane.
The IUPAC name of the alkyl halide shown is cis- 1- chloro-3-methylcyclohexane.
b)
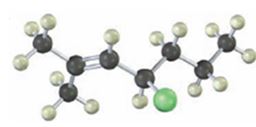
Interpretation:
The IUPAC name of the alkyl halide is to be given.
Concept introduction:
The longest continuous carbon chain in the molecule is chosen. The chain is numbered from the end which gives lowest number to the substituent either halo or alkyl group present. If different halogens are present they are numbered and listed in the alphabetical order while writing the name. If same two alternatives exist for different substituents, then the chain is numbered from the end that gives lowest number to the substituent that has alphabetical preference.
To give:
The IUPAC name of the alkyl halide shown.
Answer to Problem 14VC
The alkyl halide given is
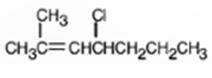
Its IUPAC name is 4-chloro-2-methyl-2-heptene.
Explanation of Solution
The compound has a seven carbon straight chain with a double bond between C2 & C3, a Cl atom on C4 and a methyl on C3. Hence its name is 4-chloro-2-methyl-2-heptene.
The IUPAC name of the alkyl halide shown is 4-chloro-2-methyl-2-heptene.
Want to see more full solutions like this?
Chapter 10 Solutions
Organic Chemistry
- Draw the products formed when the following alkynes are treated with each set of reagents: [1] H2O, H2SO4, HgSO4; or [2] R2BH followed by H2O2, −OH.arrow_forwardDraw the constitutional isomer formed when the following alkenes are treated with each set of reagents: [1] H2O, H2SO4; or [2] BH3 followed by H2O2, −OH.arrow_forwardGive IUPAC names for the following compounds:arrow_forward
- CH3 H,C-C=C-C-CH3 H2 ČH3 How to give an alkene compound according to IUPACarrow_forwardWrite down the common (not IUPAC) names of the organic molecules that would be released if this molecule were hydrolyzed: CH2−O−C—(CH2);–CH=CH–CH2–CH=CH—(CH2)4—CH3 CH-O-C-(CH2)14-CH3 O 11 CH2−O−C— (CH2)14 — CH3 Separate each name with a comma. You will find useful information in the ALEKS Data resource. 1 010 Continue O a X 000 Y F8 F9 Submiarrow_forward║? What is the structure of the alkene that would produce CH3─ C─ CH2CH3 and H─C─H when treated with ozone and then with zinc and acetic acid? ║ Oarrow_forward
- Name the major organic product formed when the following alkene undergoes a reaction with HI (Hydrohalogentation): Yarrow_forwardWrite down the common (not IUPAC) names of the organic molecules that would be released if this molecule were hydrolyzed: CH,—O—C—(CH2)—CH=CH-CH,—CH=CH—(CH2) — CH3 CH-0- -(CH2)–CH=CH(CH2)CH3 O CH,—O-C=(CH2)=CH=CH(CH2)—CH3 Separate each name with a comma and a space. You will find useful information in the ALEKS Data resource. type your answer...arrow_forwardWhat is the IUPAC name of the following compound? * CEN H2 | H2 H3C-CEC -С—с—с—ОН H3C 2-Methyl-2-hydroxymethylhex-4-yne nitrile 2-Cyano-2-methylhex-4-yne-ol 2-Hydroxymethyl-2-methylhex-4-yne nitrile 1-Cyano-2-hydroxymethyl-2-methylhex-4- ynearrow_forward
- Draw the constitutional isomer formed when the attached alkenes are treated with each set of reagents: [1] H2O, H2SO4; or [2] BH3 followed by H2O2, −OH.arrow_forwardGive IUPAC names for the following alkenes:arrow_forwardDraw the products formed when alkene reacts with these reagents: (i) H2 in the presence of Pd catalyst, (ii) HCl.arrow_forward
