
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 10.2, Problem 3LTS
Interpretation Introduction
Interpretation:
The appropriate fishhook arrows for the following radical process need to be drawn:
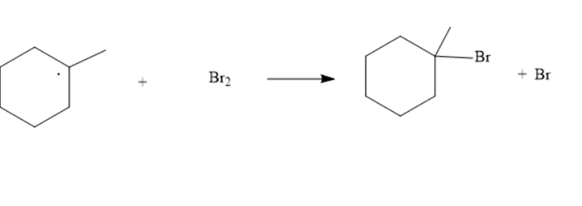
Concept Introduction :
When the bonding cannot be represented by a single Lewis formula, resonance is a means to describe delocalized electrons inside specific molecules or polyatomic ions. With such delocalized electrons, a molecule or ion can be represented by a number of resonance structures.
A radical substitution reaction is one in which one or more of the atoms or groups present in the substrate are replaced by new atoms or groups using a free radical mechanism. A radical chain reaction's initiation step is the point at which a free radical is created for the first time.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the appropriate fishhook arrows for the given radical process. Be sure to select the
fishhook arrow tool, to move a single electron.
R-
R
*O:
Edit Drawing
-R
In the substitution reaction: The product(s) of the reaction is(are)? Select all that are correct.
4
VI. Draw the each step of the mechanism for free-chain radical reaction of butane
(include the primary and secondary positions).
Chapter 10 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Give the curved arrow mechanism showing the homolysis of the appropriate C–H bond by reaction with a Br• radical. Draw curved arrows to show C–H bond homolysis. Add or remove any bonds needed.arrow_forwardDraw the electron-pushing mechanism for each radical reaction below. Identify each step as initiation, propagation, or termination.arrow_forwardDraw out the mechanismarrow_forward
- Draw the structures of the missing reactants, intermediates, or products in the following mechanism. Include all lone pairs.arrow_forwardDraw all the different monochlorinated products one would be by the radical chlorination of methylcyclopentane.arrow_forwardDraw the products of the reaction shown. Electron flow is indicated with curved arrows. H₂C CI:arrow_forward
- What carbon radical is formed by homolysis of the C–Hb bond inpropylbenzene? Draw all reasonable resonance structures for thisradical.arrow_forwardWhich of the following mechanisms is consistent with the reaction profile shown above? Circle the correct mechanism and justify your choice.arrow_forwardIs this the correct mechanism? If not, what is the correct one?arrow_forward
- Draw ONLY curved arrows for each step of the following mechanism: HỘ:arrow_forwardDraw the missing curved arrow notation for the rearrangement below. v 1st attempt O Add the missing curved arrow notation.arrow_forwardChemistry draw appropriate curved arrows and transition state! thank you!!arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning