
(a)
Interpretation:
The Fischer projections for the given molecule are to be stated.
Concept introduction:
Fischer projections is a two dimensional representation of organic compounds. It was proposed to represent glucose molecules. The carbon chain is represented vertically; the hydoxy groups and hydrogen atoms are represented horizontally, according to their stereochemistry. The
Answer to Problem 24.1P
The Fischer projections for the given molecule are shown below.
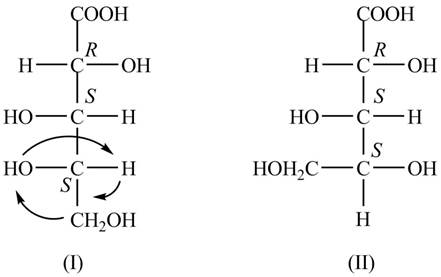
Explanation of Solution
The structure of the given molecule is shown below.
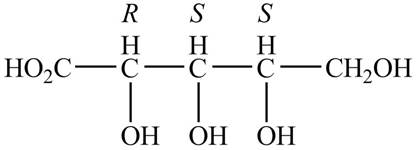
Figure 1
In the Fischer projection, the compound,
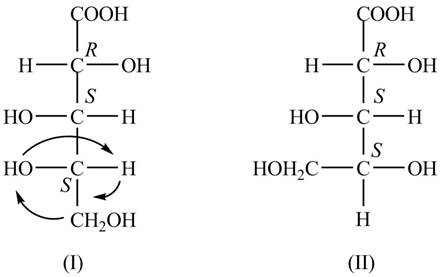
Figure 2
The Fischer projections of
(b)
Interpretation:
The Fischer projections for
Concept introduction:
Fischer projections is a two dimensional representation of organic compounds. It was proposed to represent glucose molecules. The carbon chain is represented vertically; the hydoxy groups and hydrogen atoms are represented horizontally, according to their stereochemistry. The aldehyde group is paced at the top of the carbon chain and numbered
Answer to Problem 24.1P
The Fischer projections for
Explanation of Solution
The structure of
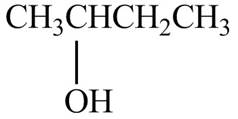
Figure 3
In the Fischer projection, the compound,
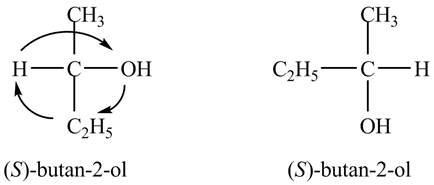
Figure 4
The Fischer projections of
Want to see more full solutions like this?
Chapter 24 Solutions
Organic Chemistry
- Write each of the following condensed structural formulas as a bond-line formula (that is, using only lines and no elemental symbols). (a) CH3CH2CH2CH2CH3 (b) (CH3)2CHCH2CH3arrow_forwardExplain why each of the following alkenes is stable or unstable.(a) 1,2-dimethylcyclopentene (b) trans-1,2-dimethylcyclopentene(c) trans-3,4-dimethylcyclopentenearrow_forward4. (a) Draw a skeletal (line-bond) structure for 3,4-dimethylhexane. (b) Draw a sawhorse representation of any staggered conformation of this molecule looking down the carbon-3 to carbon-4 bond. (c) Draw a Newman projection looking down the carbon-3 to carbon-4 bond of the same conformation that you drew as a sawhorse representation.arrow_forward
- 4. Which of the following has a higher boiling point? Explain your answer:(a) 2-propanol or 2-butanol(b) Dimethyl ether or ethanolarrow_forward2. Draw one enol tautomer of each of the following ketones and aldehydes. a) b) c) of مل G Harrow_forward(a) which if the structure of trans-1,2-dimethylcyclopentane? (b) which is the most stable conformation of 1-bromo-2-ethylcyclohexane? (c) which is the least stable conformation of 1-bromo-2-ethylcyclohexane? (d) which is the more stable configuration of 1,3-dimethylcyclopentane? *Et = ethylarrow_forward
- Arrange the alkanes in each set in order of increasing boiling point.(a) 2-Methylbutane, 2,2-dimethylpropane, and pentane(b) 3,3-Dimethylheptane, 2,2,4-trimethylhexane, and nonanearrow_forwardDraw the structures that correspond to their IUPAC or common name. (c) 4-mercapto-1-butanol (d) trans-1,3-diethoxycyclopentanearrow_forward(a)({)-hexane-3,4-diol, label each structure you have drawn as chiral or achiral.arrow_forward
- Write the correct IUPAC name for each of the following. (a) CH3 i (b) (c) CH3CH₂CH- 요 CH3—O—CH,CH,CH,CH2 -C-OH CH3 C-OH CH3CHCHCH₂-C-OH CH3arrow_forwardDraw the following compound.(2E)-but-2-enal (3Z)-4-hydroxypent-3-enoic acidarrow_forwardDraw the structure of all compounds that fit the following descriptions. nine constitutional isomers having the molecular formula C7H16arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





