
Concept explainers
(a)
Interpretation: The major product of the following reaction is to be interpreted.
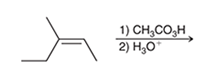
Concept introduction:
The dihydroxylation reaction of
(b)
Interpretation: The major product of the following reaction is to be interpreted.
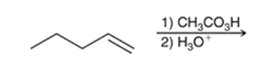
Concept introduction:
The dihydroxylation reaction of alkene involves the addition of
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
ORGANIC CHEMISTRY
- Provide the major products (write “no reaction” if you think so) for the following reactions with correct stereochemistry.arrow_forwardShow how the following starting materials are converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.arrow_forwardPredict the product(s) and provide the mechanism for each reaction below.arrow_forward
- 5) Predict the product and provide a mechanism and stereochemistry of the following reaction. 0 H₂SO4 + CH3OH- 1arrow_forwardIdentify the expected major product of the following Cope rearrangement. A II IV O II O Varrow_forwardPlease provide the intermediates, steps, and labels. Thanks!arrow_forward
- Which of the ketones below could not be prepared by an acid-catalyzed hydration of an alkyne?arrow_forwardGive me a clear handwritten Detailed Solutionarrow_forwardPredict the major organic product of each of the following reactions or provide the reagent needed to complete each transformation.arrow_forward
- Predict all possible products for the following reactions: please clearly show MAJOR product, MINOR product, and TRACE product (if it is possible theoretically but not expected to be observed). If no products are expected, please say NO REACTION: ? O₂N A NO₂ NO₂ HNO3 NO₂ H₂SO4 B NO₂ NO₂ O₂N C NO₂ D NO REACTIONarrow_forwardExplain in detail which of the following compounds (A or B) when reacted with acetophenone under basic conditions will undergo a 1,4 addition reaction. Predict the product of the reaction and propose a mechanism.arrow_forwardElimination, stereochemical aspects. Draw the major product that is obtained when (2R,3R)-2-Bromo-3-phenylbutane is treated with sodium ethoxide and explain the stereochemical outcome of the reaction using Newman projections.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

