
CHEMISTRY-MOD.MASTERING (18W)
8th Edition
ISBN: 9780136780922
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 7.81SP
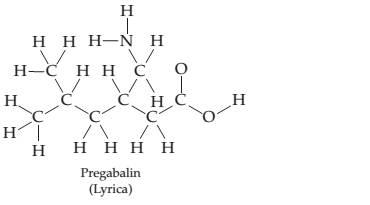
Pregabalin
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Designate the structural features of polar molecules using partial charges or arrows.
Ethyl heptanoate is responsible for grape aroma. It has the chemical formula C9H1802 but structural
formula CH3-(CH2)s-COO-CH2-CH3. A structure is shown below. Which bonds would feature partial
charges? Designate them using partial charges or arrows.
H
H
H
| H
H
H
H
Histamine (C, H, N,) is responsible for runny nose and watery eyes of allergies, and it stimulates the secretion of stomach acid. Fill in all lone pairs and double
bonds to give every atom its usual bonding pattern.
XHr
HHH
13
:0
th
Sort the following
molecules according to
increasing bond angles
about the central atom:
N3, BCI3, NH3, BrCl5.
Chapter 7 Solutions
CHEMISTRY-MOD.MASTERING (18W)
Ch. 7 - Use the electro negativity values in Figure 7.4...Ch. 7 - Conceptual APPLY 7.2 An electrostatic potential...Ch. 7 - The dipole moment of AgCI in the gas phaseis...Ch. 7 - Predict which bond has greater percent ionic...Ch. 7 - Select the correct electron-dot structure for H2S...Ch. 7 - Use the octet rule to predict the molecular...Ch. 7 - Identify the correct electron-dot structure for...Ch. 7 - Draw an electron-dot structure for each of the...Ch. 7 - Select the correct electron-dot structure for...Ch. 7 - Identify the correct electron-dot structure(s) for...
Ch. 7 - Prob. 7.11PCh. 7 - Which oxygen species do you predict to be most...Ch. 7 - Draw an electron-dot structure for the following...Ch. 7 - There are two molecules with the formula C2H6O...Ch. 7 - The following structure is a representation of...Ch. 7 - Draw two possible electron-dot structures for the...Ch. 7 - Called “laughing gas, nitrous oxide (N2O) is...Ch. 7 - Draw as many resonance structures as possible for...Ch. 7 - Prob. 7.19PCh. 7 - Prob. 7.20ACh. 7 - Calculate the formal charge on each atom in the...Ch. 7 - Start with the electron-dot structure for the...Ch. 7 - Calculate formal charges on the C and O atoms in...Ch. 7 - Three resonance structures for anisole (Problem...Ch. 7 - The toxicity of the organophosphate insecticides...Ch. 7 - Prob. 7.26PCh. 7 - Prob. 7.27PCh. 7 - The following structure is a representation of the...Ch. 7 - The electron-dot structure for the nerve a gentsar...Ch. 7 - Draw the new electron-dot structures indicated by...Ch. 7 - The following diagram shows the potential energy...Ch. 7 - The following diagram shows the potential energy...Ch. 7 - Two electrostatic potential maps are shown, one of...Ch. 7 - Prob. 7.34CPCh. 7 - Which of the following drawings is most likely to...Ch. 7 - The following ball-and-stick molecular model is a...Ch. 7 - The following hall-and-stick molecular model is a...Ch. 7 - Sinapaldehyde, a compound present in the toasted...Ch. 7 - Vitamin C (ascorbic acid) has the following...Ch. 7 - Match the following descriptions with the type of...Ch. 7 - Why do two atoms come together to form a covalent...Ch. 7 - Explain the difference in the bond dissociation...Ch. 7 - Explain the difference in the bond dissociation...Ch. 7 - Predict which of the following bonds should be...Ch. 7 - Prob. 7.45SPCh. 7 - What general trends in electro negativity occur in...Ch. 7 - Predict the electro negativity of the undiscovered...Ch. 7 - Order the following elements according to...Ch. 7 - Order the following elements according to...Ch. 7 - Which of the following substances contain bonds...Ch. 7 - Use the electro negativity data in Figure 7.4 to...Ch. 7 - Show the direction of polarity for each of the...Ch. 7 - Show the direction of polarity for each of the...Ch. 7 - Which of the substances...Ch. 7 - Which of the substances...Ch. 7 - Order the following compounds according to the...Ch. 7 - Order the following compounds according to the...Ch. 7 - Prob. 7.58SPCh. 7 - Using only the elements Ca, Cl, and Si, give...Ch. 7 - The dipole moment of BrCl is 0.518 D, and the...Ch. 7 - Prob. 7.61SPCh. 7 - Prob. 7.62SPCh. 7 - Prob. 7.63SPCh. 7 - Why does the octet rule apply primarily to...Ch. 7 - Which of the following substances contains an atom...Ch. 7 - Draw electron-dot structures for the following...Ch. 7 - Draw electron-dot structures for the following...Ch. 7 - Identify the correct electron-dot structure for...Ch. 7 - Draw an electron.dot structure for the hydronium...Ch. 7 - Oxalic acid, H2C2O4 , is a mildly poisonous...Ch. 7 - Draw an electron-dot structure for carbon...Ch. 7 - Prob. 7.72SPCh. 7 - Identify the fourth-row elements, X, that form the...Ch. 7 - Write electron-dot structures for molecules with...Ch. 7 - Write electron-dot structures for molecules with...Ch. 7 - Which compound do you expect to have the stronger...Ch. 7 - Which compound do you expect to have the stronger...Ch. 7 - Draw an electron-dot structure for each of the...Ch. 7 - Prob. 7.79SPCh. 7 - Methylphenidate (C14H19NO2) , marketed as Ritalin,...Ch. 7 - Pregabalin (C8H17NO2) , marketed as Lyric a, is an...Ch. 7 - The following molecular model is that of...Ch. 7 - Ibuprofen C 13 H 18 O 2 marketed under such brand...Ch. 7 - Draw as many resonance structures as you can that...Ch. 7 - Draw as many resonance structures as you can for...Ch. 7 - Which of the following pairs of structures...Ch. 7 - Which of the following pairs of structures...Ch. 7 - Draw as many resonance structures as you can that...Ch. 7 - Draw as many resonance structures as you can that...Ch. 7 - Benzene has the following structural formula. Use...Ch. 7 - Draw three resonance structures for sulfur...Ch. 7 - Some mothballs used when storing clothes are made...Ch. 7 - Four different structures (a), (b), (c), and (d)...Ch. 7 - Draw an electron-dot structure for carbon...Ch. 7 - Assign formal charges to the atoms in the...Ch. 7 - Assign formal charges to the atoms in the...Ch. 7 - Assign formal charges to the atoms in the...Ch. 7 - Assign formal charges to the atoms in the...Ch. 7 - Calculate formal charges for the C and O atoms in...Ch. 7 - Draw two electron-dot resonance structures that...Ch. 7 - Draw two electron-dot resonance structures that...Ch. 7 - Prob. 7.102SPCh. 7 - Prob. 7.103SPCh. 7 - Boron trifluoride reacts with dimethyl ether to...Ch. 7 - Thiofulminic acid, HCNS, has recently been...Ch. 7 - Draw two rcsonancc strutur for methyl isocyanate,...Ch. 7 - In the cyanatc ion. OCN , carbon is the central...Ch. 7 - Prob. 7.108MPCh. 7 - Prob. 7.109MPCh. 7 - Prob. 7.110MPCh. 7 - The neutral OH molecule has been implicated in...Ch. 7 - Prob. 7.112MPCh. 7 - Prob. 7.113MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Following are the structures of three isomers of difluorobenzene, C6H4F2. Are any of them nonpolar?arrow_forwardIndicate which of the following molecules are polar. Draw the molecular structure of each polar molecule, including the arrows that indicate the bond dipoles and the molecular dipole moment. (a) HCN (b) I2 (c) NOarrow_forwardHistidine is an essential amino acid that the body uses to form proteins. The Lewis structure of histidine follows. What are the approximate values for bond angles 1 through 5 (indicated on the structure by blue numbers)?arrow_forward
- Formamide, HC(O)NH2, is prepared at high pressures from carbon monoxide and ammonia, and serves as an industrial solvent (the parentheses around the O indicate that it is bonded only to the carbon atom and that the carbon atom is also bonded to the H and the N atoms). Two resonance forms (one with formal charges) can be written for formamide. Write both resonance structures, and predict the bond angles about the carbon and nitrogen atoms for each resonance form. Are they the same? Describe how the experimental determination of the HNH bond angle could be used to indicate which resonance form is more important.arrow_forwardIn addition to CO, CO2, and C3O2, there is another molecular oxide of carbon, pentacarbon dioxide, C5O2, a yellow solid. (a) What is the approximate C-to-C-to-O bond angle in pentacarbon dioxide? (b) What is the approximate C-to-C-to-C bond angle in this compound?arrow_forwardlo Bond energies energy (kJ/mol) 413 460 350 293 211 370 602 839 385 532 1077 356 615 bond C-H C-F C-CI C-Br C-I C-C C=C C=C C-O C=0 CEO C-N C=N CEN C-P C-S C=S C-Si 749 263 312 397 375 bond N-H N-C N=C N=C N-O N=O N=O N-N N=N N=N N-P N-F N-CI N-Br energy (kJ/mol) 391 356 615 749 206 631 1020 277 418 945 209 349 334 281 bond S-H S-F S-CI S-Br S-I S-S S=S energy (kJ/mol) 381 392 272 230 206 271 418 Bond energies bond H-H H-F H-CI H-Br H-I H-C H-O H-N H-P H-S energy (kJ/mol) 436 570 431 366 298 413 497 391 351 381 bond O-H O-F O-CI O-Br O-I O-C O=C O=C 0-0 0=0 O-N O=N O=S energy (kJ/mol) 497 215 234 210 213 385 532 1077 210 498 206 631 551 bond F-F F-CI F-Br F-I CI-CI Cl-Br CI-I Br-Br Br-I I-I energy (kJ/mol) 159 261 280 272 243 219 211 194 179 152 Using the appropriate bond energies, calculate the heat of reaction AH for the following reaction: H-H+CI-CI-2 H-CI You can find a table of bond energies by using the Data button on the ALEKS toolbar. Round your answer to the nearest kJ/mol.…arrow_forward
- The following molecule is a derivative of moclobemide, a drug used to treat anxiety and depression. CI The approximate C-C-CI bond angle indicated with the star is: O 79° O 120 O 109.5° O 90° O 180°arrow_forwardGlue adhesion usually requires a dry surface, so studying the chemistry behind the attachment of mussels to wet rocks can lead to the development of new adhesive materials. Chemists used the compound shown to model the mussel foot proteins involved in the adhesion process (Science 2015, 349, 628-632). Some of the nitrogen atoms in this structure are believed to play a critical role in achieving the strong binding. Identify the number of lone pairs on each nitrogen atom in the following structure. но но 8 H N° NH3 10 "Ne 6. タール OH 3. HO HO. 1 N: 6 N: 2 N: 7 N:arrow_forwardDraw a three dimensional model of methanol, CH3OH, including lone pairs of electrons using wedges, dashes and straight linesarrow_forward
- do bond angte 7A 6A Molecular Geometry Chart 5A 4A ЗА 1A 2A HF ROUP NO: H,O NH, CH, RIOD 2 drides BH, 'LiH BeH2 ewis Diagram tructural Diagram #Shared e-pairs on central atom #Unshared e-pairs on central atom Bond angle Molecular Shape:arrow_forwardPolarity (or ion) Formula Lewis structure AXE Electron group Molecular Bond Notation geometry geometry angle PF3 H20 PF5 SF4arrow_forwardThe SeF6 molecule displays which bond angles (in degrees)about the central Se? VSEPR theory predicts which molecular structure (arrangement of bonded atoms about the central Se atom) for SeF6?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY