
Classify each
(a)
Interpretation:
To classify the given alkyl halide as 1°, 2°, or 3°
Concept introduction:
Alkyl halides are organic molecules that contains a halogen atom X bonded to sp3 hybridized carbon atom. Alkyl halides are classified as primary (1°), secondary (2°), and tertiary (3°) depending on the number of carbons bonded to the carbon with the halogen.
Answer to Problem 7.1P
CH3CH2CH2CH2CH2-Br---➜ primary (1°)
Explanation of Solution
Alkyl halides are classified as primary (1°), secondary (2°), and tertiary (3°) depending on the number of carbons bonded to the carbon with the halogen.
primary (1°) = one carbon attached to carbon with halogen
secondary (2°) = two carbon attached to carbon with halogen
tertiary (3°) = three carbon attached to carbon with halogen
CH3CH2CH2CH2CH2-Br---➜ in this structure only one carbon attached to carbon with halogen so primary (1°)
Thus the given structure is primary (1°)
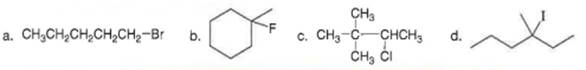
(b)
Interpretation:
To classify the given alkyl halide as 1°, 2°, or 3°
Concept introduction:
Alkyl halides are organic molecules that contains a halogen atom X bonded to sp3 hybridized carbon atom. Alkyl halides are classified as primary (1°), secondary (2°), and tertiary (3°) depending on the number of carbons bonded to the carbon with the halogen.
Answer to Problem 7.1P
 ------------➜ tertiary (3°)
------------➜ tertiary (3°)
Explanation of Solution
Alkyl halides are classified as primary (1°), secondary (2°), and tertiary (3°) depending on the number of carbons bonded to the carbon with the halogen.
primary (1°) = one carbon attached to carbon with halogen
secondary (2°) = two carbon attached to carbon with halogen
tertiary (3°) = three carbon attached to carbon with halogen
 ---➜ in this structure three carbons attached to carbon with halogen so primary (1°)
---➜ in this structure three carbons attached to carbon with halogen so primary (1°)
Thus the given structure is tertiary (3°)
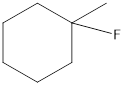
(c)
Interpretation:
To classify the given alkyl halide as 1°, 2°, or 3°
Concept introduction:
Alkyl halides are organic molecules that contains a halogen atom X bonded to sp3 hybridized carbon atom. Alkyl halides are classified as primary (1°), secondary (2°), and tertiary (3°) depending on the number of carbons bonded to the carbon with the halogen.
Answer to Problem 7.1P
 ------------➜ secondary (2°)
------------➜ secondary (2°)
Explanation of Solution
Alkyl halides are classified as primary (1°), secondary (2°), and tertiary (3°) depending on the number of carbons bonded to the carbon with the halogen.
primary (1°) = one carbon attached to carbon with halogen
secondary (2°) = two carbon attached to carbon with halogen
tertiary (3°) = three carbon attached to carbon with halogen

---➜ in this structure two carbons attached to carbon with halogen so secondary (2°)
Thus the given structure is secondary (2°)
(d)
Interpretation:
To classify the given alkyl halide as 1°, 2°, or 3°
Concept introduction:
Alkyl halides are organic molecules that contains a halogen atom X bonded to sp3 hybridized carbon atom. Alkyl halides are classified as primary (1°), secondary (2°), and tertiary (3°) depending on the number of carbons bonded to the carbon with the halogen.
Answer to Problem 7.1P
![]() ------------➜ tertiary (3°)
------------➜ tertiary (3°)
Explanation of Solution
Alkyl halides are classified as primary (1°), secondary (2°), and tertiary (3°) depending on the number of carbons bonded to the carbon with the halogen.
primary (1°) = one carbon attached to carbon with halogen
secondary (2°) = two carbon attached to carbon with halogen
tertiary (3°) = three carbon attached to carbon with halogen
![]()
---➜ in this structure three carbons attached to carbon with halogen so tertiary (3°)
Thus the given structure is tertiary (3°)
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry
- Identify the most important aldehyde and ketone from Section 14.4 on the basis of amount used, and list at least one characteristic for each that contributes to its usefulness.arrow_forwardArrange these compounds in order of increasing boiling point. (a) 1-butanol, butane, diethylether (b) hexane, 1-hexanol, dipropyletherarrow_forwardH3C. H3C CH3 Br 1-propanolarrow_forward
- Name each aldehyde or ketone. CH, CH, c. CH3-C-CH-CH-CH-C-H ČH,arrow_forwardGive the structure corresponding to each IUPAC name. 2,4 dimethyl- 2 hexanolarrow_forwardOrganic Reaction Write an equation for the oxidation of each alcohol. Use [O] above the arrow to indicate an oxidizing agent. If no reaction occurs, write "no reaction" after the arrow. 1. CH3CH2CH2CH2CH2OH CH₂ CH₂CCH₂CH₂ ОН CH CHCH CH CH CH, ОНarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning



