
(a)
Interpretation:
The stereoisomer products for the given reaction should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Electrophilic addition: It is a type of addition reaction in which the pi bond present in the molecule breaks as the electrophile approaches and results in the formation of product with sigma bond.
In addition reaction of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
E configuration: The geometric isomers are given E configuration if high priority groups are placed on opposite sides of the bond.
Z configuration: The geometric isomers are given Z configuration if high priority groups are placed on same sides of the bond.
Stereo specific: The reaction is considered as stereo specific if the reactant is stereo isomers that give rise to different set of stereo isomers.
Stereoisomers: Two compounds with same molecular formula but different in their orientation are considered as isomers.
The presence of atom with non-super impossible mirror image is defined as enantiomers which are given
(b)
Interpretation:
The stereoisomer products for the given reaction should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Electrophilic addition: It is a type of addition reaction in which the pi bond present in the molecule breaks as the electrophile approaches and results in the formation of product with sigma bond.
In addition reaction of alkenes when two substituents approaches same side of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
E configuration: The geometric isomers are given E configuration if high priority groups are placed on opposite sides of the bond.
Z configuration: The geometric isomers are given Z configuration if high priority groups are placed on same sides of the bond.
Stereo specific: The reaction is considered as stereo specific if the reactant is stereo isomers that give rise to different set of stereo isomers.
Stereoisomers: Two compounds with same molecular formula but different in their orientation are considered as isomers.
The presence of atom with non-super impossible mirror image is defined as enantiomers which are given
(c)
Interpretation:
The stereoisomer products for the given reaction should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Electrophilic addition: It is a type of addition reaction in which the pi bond present in the molecule breaks as the electrophile approaches and results in the formation of product with sigma bond.
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Acid Catalyzed Hydration Reaction: The reaction involves breaking of phi bonds between carbon-carbon multiple bonds and addition of alcohol to more substituted position of carbon in the molecule.
First step is the acid donates proton to the alkene which leads to the formation of more stable carbo cation.
Then, the water is added to the given alkene through acid catalyzed reaction where the water gets added to the carbo cation finally, the removal of one proton from oxonium ion (oxygen with one positive charge) using water results in the formation of product.
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:
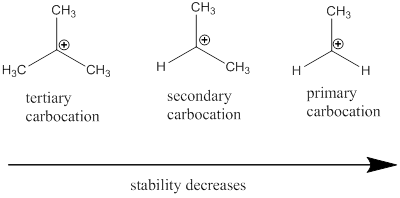
(d)
Interpretation:
The stereoisomer products for the given reaction should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Electrophilic addition: It is a type of addition reaction in which the pi bond present in the molecule breaks as the electrophile approaches and results in the formation of product with sigma bond.
In addition reaction of alkenes when two substituents approaches same side of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
E configuration: The geometric isomers are given E configuration if high priority groups are placed on opposite sides of the bond.
Z configuration: The geometric isomers are given Z configuration if high priority groups are placed on same sides of the bond.
Stereo specific: The reaction is considered as stereo specific if the reactant is stereo isomers that give rise to different set of stereo isomers.
Stereoisomers: Two compounds with same molecular formula but different in their orientation are considered as isomers.
The presence of atom with non-super impossible mirror image is defined as enantiomers which are given
(e)
Interpretation:
The stereoisomer products for the given reaction should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Electrophilic addition: It is a type of addition reaction in which the pi bond present in the molecule breaks as the electrophile approaches and results in the formation of product with sigma bond.
In addition reaction of alkenes when two substituents approaches same side of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
E configuration: The geometric isomers are given E configuration if high priority groups are placed on opposite sides of the bond.
Z configuration: The geometric isomers are given Z configuration if high priority groups are placed on same sides of the bond.
Stereo specific: The reaction is considered as stereo specific if the reactant is stereo isomers that give rise to different set of stereo isomers.
Stereoisomers: Two compounds with same molecular formula but different in their orientation are considered as isomers.
The presence of atom with non-super impossible mirror image is defined as enantiomers which are given
(f)
Interpretation:
The stereoisomer products for the given reaction should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Electrophilic addition: It is a type of addition reaction in which the pi bond present in the molecule breaks as the electrophile approaches and results in the formation of product with sigma bond.
In addition reaction of alkenes when two substituents approaches same side of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
E configuration: The geometric isomers are given E configuration if high priority groups are placed on opposite sides of the bond.
Z configuration: The geometric isomers are given Z configuration if high priority groups are placed on same sides of the bond.
Stereo specific: The reaction is considered as stereo specific if the reactant is stereo isomers that give rise to different set of stereo isomers.
Stereoisomers: Two compounds with same molecular formula but different in their orientation are considered as isomers.
The presence of atom with non-super impossible mirror image is defined as enantiomers which are given
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
EBK ORGANIC CHEMISTRY
- h. How do you convert 1,4 -Dibromo-2-butyne to Cis-1,4 -Dibromo-2-butene? i. How do you convert 1,4 -Dibromo-2-butyne to Trans-1,4 -Dibromo-2-butene?arrow_forwardName the following compound: O A. 3-methoxyhex-3-ene O B. (Z)-4-methoxyhex-4-ene O C. (E)-3-methoxyhex-3-ene O D. (cis)-3-methoxyhex-3-ene O E. (Z)-3-methoxyhex-3-enearrow_forwardwhat is the starting material using Diekmann cyclization reactionarrow_forward
- g. cis-3-methylpent-2-ene with BH3, H2O2, and OH CH3 H3C- CH =C -CH2-CH3 Major Product(s) of g. Stereochemistry of the product described in g. (Circle one) а. R b. S С. Е d. Z e. a mixture of E and Z f. erythro g. threo h. a racemic (50/50) mixture i. all 4 products (a set of erythro and a set of threo) j. no stereochemistryarrow_forwardh. 3-methyl-1-butene with Hg(OAC)2 and CH3OH, followed later by NaBH4 CH3 H2C=CH-CH-CH3 Major Product(s) of h. Stereochemistry of the product described in h. (Circle one) а. R b. S С. Е d. Z e. a mixture of E and Z f. erythro g. threo h. a racemic (50/50) mixture i. all 4 products (a set of erythro and a set of threo) j. no stereochemistryarrow_forward2. Consijder the reactiojn scheme below a. Draw the major product of this reaction. Show clearly (using wedge-and-dash lines) all stereoisomers formed under these reaction conditions. 1. BH3:THF 2. H2O2, NaOH b. What is the stereochemical relationship between the different stereoisomer products you drew in part 2a.?arrow_forward
- Which of the following is meso compound? O O a. (S,S)-1,3-dichlorocyclpentane b. cis-1,2-dimethylcyclohexane c. (R,R)-2,3-dibromobutane d. trans-1,2-dimethylcyclohexanearrow_forwardH3C₂ Br H₂O a. Enantiopure (1R, 3R)-3-Methylcyclohexanol b. Achiral 1-Methylcyclohexanol C. Racemic mixture (1R, 3R)- and (1S, 3S)-3-Methylcyclohexanol d. Diastereomeric mixture of (1R, 3R)- and (1S, 3R)-3-Methycyclohexanolarrow_forwardWhat is the major product for the following reaction? a. O3, CH;Cl2, -78 °C b. Me,S a coM + CO: CO2H + CO, он b + enantiomer + HCHO сно darrow_forward
- 115. Draw the products of each of the following S,2/E2 reactions. If the products can exist as stereoisomers, show which stereoisomers are formed. a. (3S,4S)-3-bromo-4-methylhexane + CH;O b. (3R,4R)-3-bromo-4-methylhexane + CH30 c. (3S,4R)-3-bromo-4-methylhexane + CH;O¯ d. (3R,4S)-3-bromo-4-methylhexane + CH3Oarrow_forwardGive the structure corresponding to each name.a. 3-bromo-4-ethylheptaneb. 1,1-dichloro-2-methylcyclohexanec. 1-bromo-4-ethyl-3-uorooctaned. (S)-3-iodo-2-methylnonanee. (1R,2R)-trans-1-bromo-2-chlorocyclohexanef. (R)-4,4,5-trichloro-3,3-dimethyldecanearrow_forwardDraw the substitution and elimination products for the following reactions, showing the configuration of each product: a. trans-1-chloro-2-methylcyclohexane + CH3O- b. cis-1-chloro-2-methylcyclohexane + CH3O− c. 1-chloro-1-methylcyclohexane + CH3O d. 1-chloro-1-methylcyclohexane + CH3OHarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





