
In the ?nal stage of the manufacturing process fora solid organic product, the product is cleaned with liquid toluene and then dried in a process whose ?owchart is shown on the next page.“
The wet product enters the dryer at a rate of 300 lbm/ h containing 0.200 lbm toluene/lbm dry solids. A stream of nitrogen at 200°F, 1.2 atm, and containing a small amount of toluene vapor also enters the dryer. (A higher temperature would cause the product to soften and degrade.) Heat is transferred in the dryer from the gas to the wet solids, causing most of the toluene to evaporate. The ?nal product contains 0.020 lbm toluene/lbm dry solids. Gas leaves the dryer at 150°F and 1.2 atm with a relative sanitation of 70% and passes through a water-cooled condenser. Gas and liquid streams leave the condenser in equilibrium at 90°F and 1 atm. The gas is reheated to 200°F and reenters the dryer.
(a) Brie?y explain this process in your own words. In your explanation, include the purposes of the condenser and the nitrogen reheater and a likely reason that nitrogen rather than air is used as the recirculating gas. What do you suppose happens to the liquid toluene leaving the condenser?
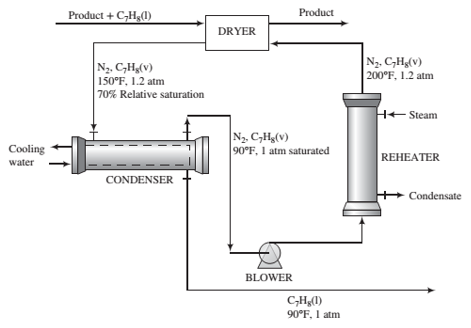
(b) Calculate the compositions (component mole fractions) of the gas streams entering and leaving the dryer, the circulation rate of dry nitrogen (lbm/h), and the volumetric ?ow rate of gas entering the dryer (ft3/h).
(c) Explain why the actual process has a small make-up nitrogen stream blended with the feed to the blower.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
ELEM.PRIN.OF CHEMICAL PROC.-W/ACCESS
Additional Engineering Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Engineering Mechanics: Dynamics (14th Edition)
Java How To Program (Early Objects)
Starting Out with Java: Early Objects (6th Edition)
Management Information Systems: Managing the Digital Firm (15th Edition)
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The





