
Custom eBook for Organic Chemistry
2nd Edition
ISBN: 9798214171104
Author: Straumanis
Publisher: Cengage Custom
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 5, Problem 25CTQ
Interpretation Introduction
Interpretation: The base with lower potential energy in bases mentioned below should be determined.
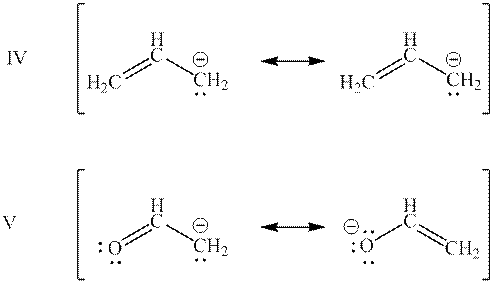
Concept introduction: When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
2. Use curved arrows to show electron movement in the reactant side and draw the product/s of the
Lewis acid-base (nucleophile-electrophile) reaction. Label the nucleophile and the electrophile. Draw in
all lone pairs and charges where appropriate.
I
Draw the outcome of the following acid-base reaction resulting from the provided curved arrows. Label the acid, base, conjugate acid, and conjugate base.
осх
(Intermediate) Reactant
ķ
H
H:
Tip: Only add curved arrows in this sketcher
1
Chapter 5 Solutions
Custom eBook for Organic Chemistry
Ch. 5 - Which elements on the periodic table (other than...Ch. 5 - You will not find “hydroxide” in the stockroom,...Ch. 5 - Prob. 3CTQCh. 5 - Prob. 4CTQCh. 5 - Prob. 5CTQCh. 5 - Prob. 6CTQCh. 5 - On which do you expect to have a more intense and...Ch. 5 - Prob. 8CTQCh. 5 - Prob. 9CTQCh. 5 - Prob. 10CTQ
Ch. 5 - Prob. 11CTQCh. 5 - Prob. 12CTQCh. 5 - Prob. 13CTQCh. 5 - Prob. 14CTQCh. 5 - Prob. 15CTQCh. 5 - Prob. 16CTQCh. 5 - For each proposed set of resonance structures: a....Ch. 5 - Consider the polarization of the C=O bond in the...Ch. 5 - The C=O double bond is called a “carbonyl bond.”...Ch. 5 - Prob. 20CTQCh. 5 - Prob. 21CTQCh. 5 - Prob. 22CTQCh. 5 - Prob. 23CTQCh. 5 - Prob. 24CTQCh. 5 - Prob. 25CTQCh. 5 - Prob. 26CTQCh. 5 - Prob. 27CTQCh. 5 - Prob. 28CTQCh. 5 - Prob. 29CTQCh. 5 - Prob. 30CTQCh. 5 - Prob. 31CTQCh. 5 - Confirm that there is no legitimate Lewis...Ch. 5 - Draw all resonance structures of the molecule...Ch. 5 - Prob. 34CTQCh. 5 - Prob. 35CTQCh. 5 - Prob. 36CTQCh. 5 - Occasionally, we will see an ionic compound that...Ch. 5 - Prob. 2ECh. 5 - Prob. 3ECh. 5 - Prob. 4ECh. 5 - Is it possible to draw a resonance structure of...Ch. 5 - Prob. 6ECh. 5 - Prob. 7ECh. 5 - Prob. 8ECh. 5 - Phenol (shown below) has a pKa10 . a. Based on pKa...Ch. 5 - Use curved arrows to show the most likely...Ch. 5 - Prob. 12ECh. 5 - Complete each Lewis structure, draw all important...Ch. 5 - Use curved arrows to show the most likely...Ch. 5 - Construct an explanation for why sulfuric acid is...Ch. 5 - Prob. 16ECh. 5 - Prob. 17ECh. 5 - Prob. 18E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For each molecule below, draw the conjugate acid or conjugate base or both if the molecule hasboth a conjugate acid and a conjugate base (e.g., water).arrow_forwardConsider the following bases: a. For each base above, circle the atom/atoms with the highest PE (will release the most P.E.when a lone pair on this atom combines with an H+ ) b. Rank the bases 1 (highest P.E./strongest base) to 7 (lowest PE/weakest base), and explainyour reasoning.arrow_forwardPlease do fastarrow_forward
- Circle the molecule that is a stronger acid. Briefly explain your choice. Use structures to support your reasoning.arrow_forwardIs this reaction overall more acidic or basic? I am a little confused because it produces an OH- but also a strong acid.arrow_forwardPlease answer 1st and 2nd with basic mechanism otherwise I'll downvotearrow_forward
- On the molecules on the back page, circle and label all of the functional groups that you see (identified in the table above). Please note the following tips that may help you on this assignment: If a molecule contains –COOH, or a C double bonded to an O and single-bonded to an –OH, the functional group you circle is only the carboxyl, not a carboxyl, carbonyl, and hydroxyl separately). Phosphate groups are tricky – any P surrounded by 4 O’s is a phosphate – adjacent phosphates may share an O, and sometimes the O is in the form of OH. There is often shorthand that is used in drawing these structures – if you see a jagged line or ring structure, assume that carbon is at each corner. There may be ionized forms of the functional groups above (such as NH2 or NH3+) – these are the same functional groups. The order of the atoms in the functional group may be switched, depending on which side of the molecule it is on. For example, “—NH3” and “H3N—“ are the same thing.arrow_forwardOn the molecules on the back page, circle and label all of the functional groups that you see (identified in the table above). Please note the following tips that may help you on this assignment: If a molecule contains –COOH, or a C double bonded to an O and single-bonded to an –OH, the functional group you circle is only the carboxyl, not a carboxyl, carbonyl, and hydroxyl separately). Phosphate groups are tricky – any P surrounded by 4 O’s is a phosphate – adjacent phosphates may share an O, and sometimes the O is in the form of OH. There is often shorthand that is used in drawing these structures – if you see a jagged line or ring structure, assume that carbon is at each corner. There may be ionized forms of the functional groups above (such as NH2 or NH3+) – these are the same functional groups. The order of the atoms in the functional group may be switched, depending on which side of the molecule it is on. For example, “—NH3” and “H3N—“ are the same thing.arrow_forwardOn the molecules on the back page, circle and label all of the functional groups that you see (identified in the table above). Please note the following tips that may help you on this assignment: If a molecule contains –COOH, or a C double bonded to an O and single-bonded to an –OH, the functional group you circle is only the carboxyl, not a carboxyl, carbonyl, and hydroxyl separately). Phosphate groups are tricky – any P surrounded by 4 O’s is a phosphate – adjacent phosphates may share an O, and sometimes the O is in the form of OH. There is often shorthand that is used in drawing these structures – if you see a jagged line or ring structure, assume that carbon is at each corner. There may be ionized forms of the functional groups above (such as NH2 or NH3+) – these are the same functional groups. The order of the atoms in the functional group may be switched, depending on which side of the molecule it is on. For example, “—NH3” and “H3N—“ are the same thing.arrow_forward
- a. Which of the following has a greater electronegativity? (circle one) Carbon / Cesium b. Explain why you chose your answer in Za.arrow_forwardSolve both parts otherwise I will downvote...arrow_forwardPrackce Compare the two hydrogens shown in this compound and circle the one that is more acidic. H-N а. Draw the two possible conjugate bases in red b. For your answer above, circle which of the conjugate bases is more stable b. Briefly justify your answer in part b (more space on next page if you need it).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning