
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3.5, Problem 3.31P
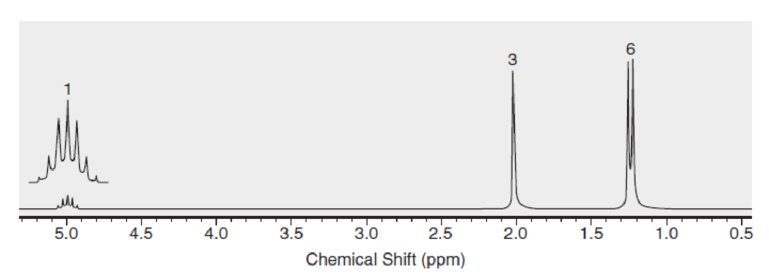
Below are NMR spectra of several compounds. Identify whether these compounds are likely to contain ethyl, isopropyl, and/or tert-butyl groups:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Give the structure for two isomers of molecular formula C4H10O which are consistent with the ^1H-NMR spectra shown below.
How many signals would you expect to see in the 13C NMR spectrum for the following compound?
Determine the structure of a compound with the formula C11H15ClO2 based on the 1H NMR spectrum below. Based on its 13C NMR (DEPT) spectrum, it is evident that this molecule shows 9 signals including one methyl (CH3) and four methylene (CH2) units.
Chapter 3 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 3.1 - Prob. 3.2PCh. 3.1 - Prob. 3.3PCh. 3.1 - Prob. 3.4PCh. 3.1 - Prob. 3.5PCh. 3.1 - Prob. 3.6PCh. 3.1 - Prob. 3.7PCh. 3.1 - Prob. 3.8PCh. 3.1 - Prob. 3.9PCh. 3.1 - Prob. 3.10PCh. 3.1 - If you look at your answers to the previous...
Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Prob. 3.19PCh. 3.3 - Prob. 3.21PCh. 3.3 - Prob. 3.22PCh. 3.3 - Prob. 3.23PCh. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.9 - Prob. 3.43PCh. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Prob. 3.48PCh. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
110. Have each group member choose a set of quantum numbers for an electron in a hydrogen atom. Calculate the w...
Chemistry: Structure and Properties (2nd Edition)
For the 3pz and 4dxz hydrogen-like atomic orbitals, sketch the following: a. The radial function R b. The radia...
Inorganic Chemistry
Two curves for the same reaction are shown in the following reaction energy diagram. Which curve represents the...
General, Organic, and Biological Chemistry (3rd Edition)
6.1 State the number of electrons that be must be lost by atoms of each of the following to achieve a stable el...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
A circular tube of diameter D=0.2mm and length L=100mm in1poes a constant heat flux of qn=20103W/m2 on a fluid ...
Fundamentals of Heat and Mass Transfer
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Explain why 2-chloropropene shows signals for three kinds of protons in its 1H NMRspectrum? Draw the structure and predict the approximate values for 1H-NMR spectra.arrow_forwardThe carbon NMR chemical shifts of diethylmethylamine, piperidine, propan-1-ol, andpropanal follow. Determine which spectrum corresponds to each structure, and showwhich carbon atom(s) are responsible for each absorption.7.9, 44.7, 201.9arrow_forwardHow could 1H NMR spectroscopy be used to distinguish between attachedpair of compoundsarrow_forward
- Predict the 1H NMR spectrum for the compound below and sketch it. Include splitting, and label the protons and their corresponding peaks.arrow_forwardFigures below give a molecular formula, an infrared spectrum,and a 1H NMR spectrum for an unknown compound.What is the structure of the compound? Explain briefly.arrow_forwardThe following is the predicted 1H-NMR spectrum for an unknown compound with molecular formula C6H14O . This compound is a liquid at room temperature, is slightly soluble in water, and reacts with sodium metal with the evolution of a gas.arrow_forward
- Organic chemistry 2: from the attached NMR spectrum what can be the proposed molecule that corresponds to the spectrum. Please discuss how how you arrived at the identity of the compound.arrow_forwardThe following NMR spectra were obtained from a compound with the molecular formula C4H6O. Use this information to predict its structure.arrow_forwardHow could 1H NMR spectroscopy be used to distinguish betweencompounds X and Y?arrow_forward
- Consider the 13C NMR spectrum below. The spectrum belongs to which one of the following constitutional isomers of the compound C9H12? Select the single best answer.arrow_forwardThe MS and 13C NMR spectra for two constitutional isomers with 4 degrees of unsaturation are shown below. Determine the molecular structures of the two constitutional isomers.arrow_forwardPredict the theoretical number of different NMR signals produced by each compound,and give approximate chemical shifts. Point out any diastereotopic relationships. cyclopentanolarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY