
Concept explainers
In the following drawings, red spheres represent cations and blue spheres represent anions. Match each of the drawings (a)–(d) with the following ionic compounds:
- (i) Ca3(PO4)2
- (ii) Li2CO3
- (iii) FeCl2
- (iv) MgSO4
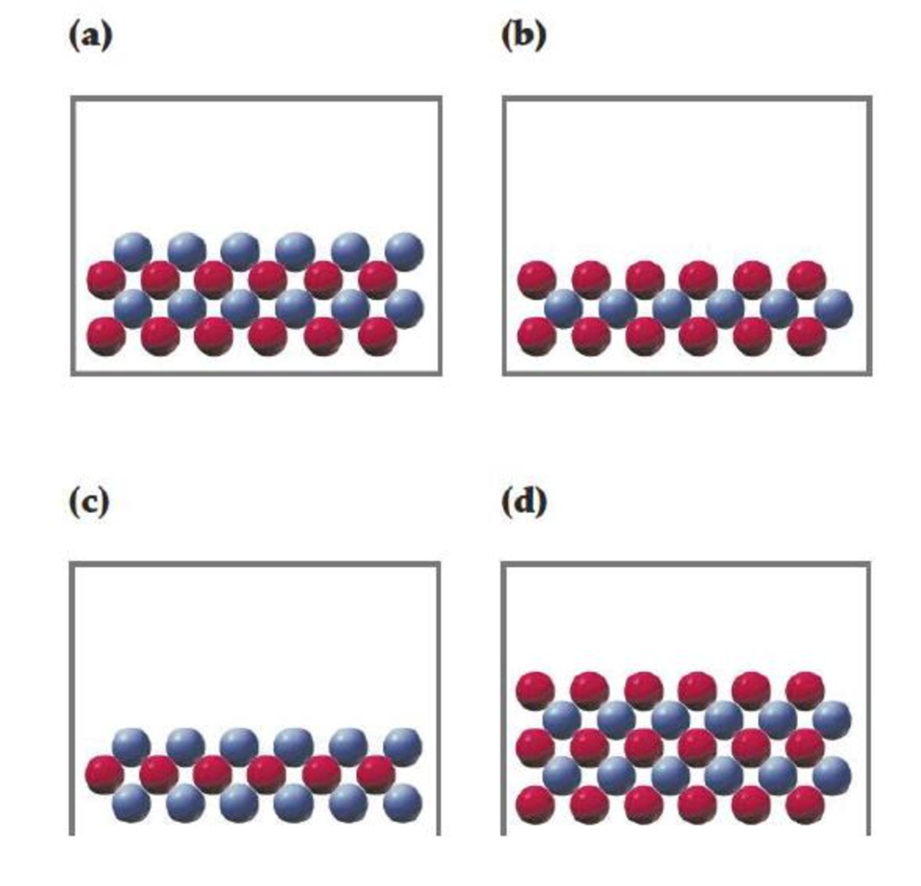
Interpretation:
For the given compounds correct representation of diagramhas to be chosen.
Explanation of Solution
To choose: Correct representation of diagram for given compounds.
Given,
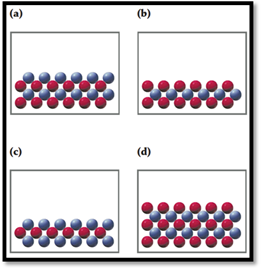
Figure 1
Consider red sphere as cation and blue sphere as anion.
The ratio of cation (
The ratio of cation (
The ratio of cation (
The ratio of cation (
For the given compounds correct representation of diagram was chosen.
Want to see more full solutions like this?
Chapter 3 Solutions
General Chemistry: Atoms First
- Which of the following binary compounds would be expected to be an ionic compound? a. Cu2O b. CO c. NaBr d. Be3P2arrow_forward2-59 You are presented with a Lewis dot structure of element X as X.. To which two groups in the Periodic Table might this element belong?arrow_forwardGive the name of the formula of elements1, Co Cl22, N2 H43, BF3 What is the meaning of isotopy?arrow_forward
- The monatomic ions of Groups 1A(1) and 7A(17) are allsingly charged. In what major way do they differ? Why?arrow_forwardthe central atom in the nitrate anion, NO3-is surrounded byarrow_forwardArsenate is a polyatomic ion with the chemical formula AsO4. If magnesium arsenate has a chemical formula of Mg3(AsO4)2, what is the charge on the arsenate ion? Explain your reasoning?arrow_forward
- What associated anion do Ag+, Fe3+, Cu2+, Ba2+, Pb2+, and Na+ have? And what is the formula of this anion? For reactions involving dilute solutions of aqueous ammonia, what is the key substance involved in the reaction?arrow_forwardWhich element is the only transition metal that has a single-letter chemical symbol that is not the first letter of the element? A. Tungsten B. Iron C. Potassium D. Vanadium Which formula correctly represents Iron (III) oxide? A. Fe2O3 B. Fe3O2 C. FeO3 D. Fe3Oarrow_forwardIodine is an essential trace element in our diet; it is needed to produce thyroid hormone. Insufficient iodine in the diet can lead to the development of a goiter, an enlargement of the thyroid gland . The addition of small amounts of iodine to table salt (iodized salt) has essentially eliminated this health concern in the United States, but as much as 40% of the world’s population is still at risk of iodine deficiency. The iodine atoms are added as anions, and each has a 1− charge and a mass number of 127. Determine the numbers of protons, neutrons, and electrons in one of these iodine anions.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





