
Concept explainers
PRACTICE PROBLEM
If we examine Table 21.1, we find that the methylphenols (cresols) are less acidic than phenol itself. For example,
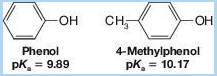
This behavior is characteristic of phenols bearing electron-releasing groups. Provide an explanation.
Interpretation:
Methyl phenols are less acidic than phenols, due to the presence of an electron releasing group on them. The statement is to be explained.
Concept introduction:
Phenols are compounds of benzene, bearing a hydroxyl group. They are alcohol derivatives but have higher acidities than alkyl alcohols.
Methyl phenols are phenol derivatives, in which a hydrogen atom attached to the carbon ring is replaced by a methyl
Answer to Problem 1PP
Solution: The
Explanation of Solution
According to table
Phenol is more acidic, as the
Want to see more full solutions like this?
Chapter 21 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry: Matter and Change
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Living by Chemistry
Living By Chemistry: First Edition Textbook
General Chemistry: Principles and Modern Applications (11th Edition)
- A) Which compounds in the picture provided has an acidic group B) Which compounds in the picture provided has a basic group? Thank You!arrow_forwardWhich best explain the basicity trends observed on compound a and b, image provided. Select all that apply. 1)Compound a is aromatic and its nitrogen’s lone pair participates in resonance. 2)Compound a is aromatic and its nitrogen’s lone pair does not participate in resonance. 3)Compound a is nonaromatic and its nitrogen’s lone pair participates in resonance. 4)Compound a is nonaromatic and its nitrogen’s lone pair does not participate in resonance. 5)Compound b is aromatic and its nitrogen’s lone pair participates in resonance. 6)Compound b is aromatic and its nitrogen’s lone pair does not participate in resonance. 7)Compound b is nonaromatic and its nitrogen’s lone pair participates in resonance. 8)Compound b is nonaromatic and its nitrogen’s lone pair does not participate in resonance.arrow_forwardanswer question and provide explanations that are easy to followarrow_forward
- e) why are they organic products in the reactions from (chemical equation for acid base reaction of benzyl alcohol and potassium hydroxide) and (Chemical equation for an acid base reaction of Ethanethiol and sodium hydroxide) more soluble in water compared to the organic reactants?arrow_forwardIs a reaction possible between an amide and alcohol? What is the product and show reaction mechanism.arrow_forwarda) Draw the mechanism for the reaction of a secondary amine with an acid chloride to make an amide.b) Explain what conditions (acid/basic/neutral; excess of one reagent, etc) are required to carry out this reaction in high yield, and why.arrow_forward
- I need help with this review question is it (A) Aromatic (B) Anti- Aromatic (C) Non- Aromaticarrow_forwardWhat is reductive amination explain with example?arrow_forward2) For two of the following pairs of compounds, identify the most basic atom in each compound, and determine which of each pair is more basic, explaining your reasoning. a) b)arrow_forward
- Have this practice synthesis problem in my textbook. I think the final solution might include the formation of a cyclic compound. Any assistance in understanding the solution to this problem would be greatly appreciated. Thanks!arrow_forwardTo practice types of aromatic reactions, this next practice problem provided us these reactions. What are the products?arrow_forwardQuestion (Organic Chemistry) Consider molecules A–B. Does the arrow pushing in each structure leadto an acceptable resonance form? If so, explain why it is acceptable or unacceptable? What are the rules and logic? If so, draw it and carefully explain your answer and provide explanations for why each structure leds to an acceptable or unacceptable resonance form. Why is the arrow pushing acceptable or unacceptable in each individual case? Explain you answer with many details, logic, and in a step-by-step fashion.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
