
CHEM: STRUC & PROP (LL) VOL 2 >C PKG<
2nd Edition
ISBN: 9780137390922
Author: Tro
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 112E
Sunscreen contains compounds that absorb ultraviolet light. When sunscreen is applied to skin, it prevents ultraviolet light from reaching the skin. The graph that follows shows the absorbance of light as a function of wavelength for two different compounds (2-EHMC and TDSA) common in sunscreen. Absorbance is a measure of the amount of light absorbed by the compound-the higher the absorbance, the more light is absorbed. Study the graph and answer the question
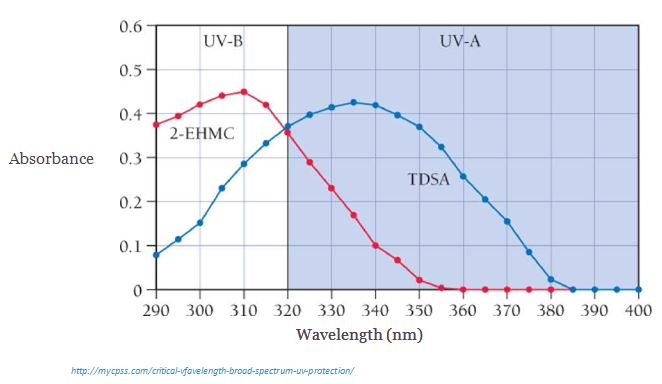
<
- Calculate the energy of a photon at the maximum absorption of TDSA.
- Calculate the energy of a photon at the maximum absorption of 2-EHMC.
- Which compound absorbs more energy at its maximum absorption?
- Why do you think sunscreens commonly contain both of these compounds and not just one of them?
- Assuming that sunlight produces 3.066 x 1022^ V ph°tQ1is, ^atthe skin absorbs one-half of these photons (and m2 • s reflects the other half), calculate the total UV energy absorbed over 0.42 m2 of skin that is exposed to sunlight for one hour. Assume that the average wavelength of the UV photons is 330 nm.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Sunscreen contains compounds that absorb ultraviolet light. Whensunscreen is applied to skin, it prevents ultraviolet light from reachingthe skin. The graph that follows shows the absorbance of light as afunction of wavelength for two different compounds (2-EHMC andTDSA) common in sunscreen. Absorbance is a measure of theamount of light absorbed by the compound—the higher theabsorbance, the more light is absorbed. Study the graph and answerthe questions below.a. Calculate the energy of a photon at the maximumabsorption of TDSA.b. Calculate the energy of a photon at the maximum absorption of 2-EHMC.c. Which compound absorbs more energy at its maximum absorption?d. Why do you think sunscreens commonly contain both of these compounds and not just one of them?e. Assuming that sunlight produces 3.066 ? 1022 ?? ?ℎ??????2?and that the skin absorbs one-half of thesephotons (and reflects the other half), calculate the total UV energy absorbed over 0.42 m2of skin thatis exposed to sunlight for…
true or false
a. Caffeine absorbs light along the visible region of the electromagnetic spectrum.
True
False
a. Particles of light emit or absorb energy with surrounding molecules. This phenomenon is governed by the Law of Conservation of Energy.
True
False
3. A prism is anle to differentiate the colors of the rainbow via refraction. The spectrometer uses a surface called a diffraction grating, which reflects light of various wavelengths at different angles. Give a real life example of an object or substance which acts as a diffracting grading, explain your reasoning.
Chapter 2 Solutions
CHEM: STRUC & PROP (LL) VOL 2 >C PKG<
Ch. 2 - Why is the quantum-mechanical model of the atom...Ch. 2 - What is light? How fast does it travel in a...Ch. 2 - Define the wavelength and amplitude of a wave.Ch. 2 - Define the frequency of electromagnetic radiation....Ch. 2 - What determines the color of light? Describe the...Ch. 2 - What determines the color of a colored object?...Ch. 2 - Give an approximate range of wavelengths for each...Ch. 2 - Explain the wave behavior known as interference....Ch. 2 - Explain the wave behavior known as diffraction....Ch. 2 - Describe the photoelectric effect. How did...
Ch. 2 - How did the photoelectric effect lead Einstein to...Ch. 2 - What is a photon? How is the energy of a photon...Ch. 2 - What is an emission spectrum? How does an emission...Ch. 2 - Describe the Bohr model for the atom. How did the...Ch. 2 - Explain electron diffraction.Ch. 2 - What is the de Broglie wavelength of an electron?...Ch. 2 - What are complementary properties? How does...Ch. 2 - Explain Heisenberg’s uncertainty principle. What...Ch. 2 - What is a trajectory? What kind of information do...Ch. 2 - Why does the uncertainty principle make it...Ch. 2 - Newton’s laws of motion are deterministic. Explain...Ch. 2 - An electron behaves in ways that are at least...Ch. 2 - What is a probability distribution map?Ch. 2 - For each solution to the Schrödinger equation,...Ch. 2 - What is a quantum-mechanical orbital?Ch. 2 - What is the Schrödinger equation? What is a wave...Ch. 2 - What are the possible values of the principal...Ch. 2 - What are the possible values of the angular...Ch. 2 - What are the possible values of the magnetic...Ch. 2 - Explain the difference between a plot showing the...Ch. 2 - Sketch the general shapes of the s, p, and d...Ch. 2 - List the four different sublevels. Given that only...Ch. 2 - Why are atoms usually portrayed as spheres when...Ch. 2 - The distance from the sun to Earth is 1.496108 km....Ch. 2 - The nearest star to our sun is Proxima Centauri,...Ch. 2 - List these types of electromagnetic radiation in...Ch. 2 - List these types of electromagnetic radiation in...Ch. 2 - Calculate the frequency of each wavelength of...Ch. 2 - Calculate the wavelength of each frequency of...Ch. 2 - Calculate the energy of a photon of...Ch. 2 - Calculate the energy of a photon of...Ch. 2 - A laser pulse with wavelength 532 nm contains 3.85...Ch. 2 - A heat lamp produces 32.8 watts of power at a...Ch. 2 - Determine the energy of 1 mol of photons for each...Ch. 2 - How much energy is contained in 1 mol of each?...Ch. 2 - Sketch the interference pattern that results from...Ch. 2 - What happens to the interference pattern described...Ch. 2 - The resolution limit of a microscope is roughly...Ch. 2 - The smallest atoms can themselves exhibit...Ch. 2 - What is the de Broglie wavelength of an electron...Ch. 2 - A proton in a linear accelerator has a de Broglie...Ch. 2 - Prob. 53ECh. 2 - A 0.22-caliber handgun fires a 27-g bullet at a...Ch. 2 - An electron has an uncertainty in its position of...Ch. 2 - An electron traveling at [1.35 \times {10^5}\] m/s...Ch. 2 - Which electron is, on average, closer to the...Ch. 2 - Which electron is, on average, further from the...Ch. 2 - What are the possible values of I for each given...Ch. 2 - What are the possible values of m1 for each given...Ch. 2 - Which set of quantum numbers cannot occur together...Ch. 2 - Which combinations of n and I represent real...Ch. 2 - Prob. 63ECh. 2 - Sketch the 3d orbitals. How do the 4d orbitals...Ch. 2 - An electron in a hydrogen atom is excited with...Ch. 2 - Determine whether each transition in the hydrogen...Ch. 2 - According to the quantum-mechanical model for the...Ch. 2 - According to the quantum-mechanical model for the...Ch. 2 - Calculate the wavelength of the light emitted when...Ch. 2 - Calculate the frequency of the light emitted when...Ch. 2 - Prob. 71ECh. 2 - An electron in a hydrogen atom relaxes to the [n =...Ch. 2 - Ultraviolet radiation and radiation of shorter...Ch. 2 - The human eye contains a molecule called...Ch. 2 - An argon ion laser puts out 5.0 W of continuous...Ch. 2 - A green leaf has a surface area of 2.50 cm2. If...Ch. 2 - Prob. 77ECh. 2 - An X-ray photon of wavelength 0.989 nm strikes a...Ch. 2 - Prob. 79ECh. 2 - The energy required to ionize sodium is 496...Ch. 2 - Prob. 81ECh. 2 - Suppose that, in an alternate universe, the...Ch. 2 - Prob. 83ECh. 2 - Prob. 84ECh. 2 - The binding energy of electrons in a metal is 193...Ch. 2 - In order for a thermonuclear fusion reaction of...Ch. 2 - Prob. 87ECh. 2 - Prob. 88ECh. 2 - The iodine molecule can be photodissociated...Ch. 2 - A sample of naphthalene in hexane contains 5.00104...Ch. 2 - Prob. 91ECh. 2 - Prob. 92ECh. 2 - Prob. 93ECh. 2 - The energy of a vibrating molecule is quantized...Ch. 2 - The wave functions for the 1s and 2s orbitals are...Ch. 2 - Before quantum mechanics was developed, Johannes...Ch. 2 - Find the velocity of an electron emitted by a...Ch. 2 - Water is exposed to infrared radiation of...Ch. 2 - Prob. 99ECh. 2 - A metal with a threshold frequency of 6.711014s1...Ch. 2 - Find the longest wavelength of a wave that can...Ch. 2 - The amount of heat to melt ice is 0.333 kJ/g. Find...Ch. 2 - Explain the difference between the Bohr model for...Ch. 2 - The light emitted from one of the following...Ch. 2 - Determine whether an interference pattern is...Ch. 2 - Which transition in the hydrogen atom results in...Ch. 2 - Discuss the nature of light with your group....Ch. 2 - How are electrons like baseballs? How are they...Ch. 2 - What are all the possible values of m1 if l = 0...Ch. 2 - Have each group member choose a set of quantum...Ch. 2 - How many nodes are there in the 1s, 2p, and 3d...Ch. 2 - Sunscreen contains compounds that absorb...Ch. 2 - Which wavelength of light has the highest...Ch. 2 - Which kind of electromagnetic radiation contains...Ch. 2 - How much energy (in J ) is contained in 1.00 mole...Ch. 2 - Light from three different lasers (A, B, and C),...Ch. 2 - Calculate the wavelength of an electron traveling...Ch. 2 - Which set of three quantum numbers does not...Ch. 2 - Calculate the wavelength of light emitted when an...Ch. 2 - Which electron transition produces light of the...Ch. 2 - How much time (in seconds) does it take light to...Ch. 2 - Which figure represents a d orbital? None of the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- It takes 348. kJ/mol to break a carbon-carbon single bond. Calculate the maximum wavelength of light for which a carbon-carbon single bond could be broken by absorbing a single photon. In nanometers (nm)arrow_forwardQ-11. Copper Sulphate (a) + Iron → Iron Sulphate (b) + Copper Write the colour of 'a' and 'b’ respectively.arrow_forwardThe absorbance of various wavelengths of light by a solution were measured. How does absorbance relate to the intensity of light that is transmitted through the solution and measured by the detector?arrow_forward
- The amount of protein in a sample is measured by the samples absorbance of light at a given wavelength. Using standards, a best fit line of absorbance vs. mg protein gave the following parameters: m = 0.01630 sm = 0.00022 b = 0.1040 sb = 0.0026 An unknown sample has an absorbance of 0.246 ± 0.0059. What is the amount of protein in the sample?arrow_forwardPlease don't provide handwritten solution ....arrow_forward2. Assuming that the lights in this room give off 572 nm, what is the frequency of the lightarrow_forward
- Chemists in the 19th century constructed an evacuated glass tube with wires inserted in both ends. When an electrical current was generated at the anode, a 'cathode ray' would pass to the cathode creating a fluorescent glow. These experiments helped chemists better understand the nature of the electron. J.J. Thomson added a magnetic field to a cathode ray tube to show that the ray would bend. His experiments demonstrated that the rays were negatively charged particles. He then calculated the mass to charge ratio of the electric ion (electron). According to his calculations, how massive was the electron relative to the hydrogen ion? Voltage Source A) The same size B) ~1000 times larger C) ~1000 times smaller + S D) ~10 times larger Anode Cathode E) ~10 times smallerarrow_forwardA carbon-oxygen double bond in a particular molecule absorbs electromagnetic radiation that has a frequency of 4.3 x 1014 s-1. What is the wavelength of this radiation in nanometers, and what type of electromagnetic radiation is this? (radio, IR, UV, visible, etc)arrow_forwardCell phone "5G" service uses a frequency of 39 GHz (39 x 109 Hz). What is the wavelenght of this radio signal? Group of answer choices 7.7 millimeters 1.5 meters 640 nanometers 3.0 kilometersarrow_forward
- Use the models to answer the following question. Model A Model B Which is the correct model and explanation for the emission of light? O A. Model A, because electrons absorb energy, release a photon of light, and jump to a higher energy level before falling back down. O B. Model A, because electrons absorb energy and jump to a higher energy level. As they fall back down that energy is released as a photon of light. O C. Model B, because electrons absorb energy, release a photon of light, and jump to a higher energy level before falling back down. O D. Model B, because electrons absorb energy and jump to a higher energy level. As they fall back down that energy is released as a photon of light. Sign out novo DII 2$ 5 e r t d f k C m altarrow_forwardHow can the periodic table be used to determine the number of valence electrons of a given element? The groups of the periodic table indicate the number of valence electrons of a given element. For example, beryllium is in the second group of the periodic table, so beryllium has two valence electrons. The number of valence electrons corresponds to the atomic number of the element. For example, helium has the atomic number of two. Therefore, helium has two valence electrons. The rows of the periodic table indicate the number of valence electrons of a given element. For example, aluminum is found in the third row of the periodic table; therefore, aluminum has three valence electrons. The periods of the periodic table indicate the number of valence electrons of a given element. For example, hydrogen is found in the first period, so hydrogen has one valence electron.arrow_forwardMuch of the evidence for the existence of atoms comes from Multiple Choice chemistry. mechanics. biology. thermodynamics. On-Screen Keyboardarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Atomic Number, Atomic Mass, and the Atomic Structure | How to Pass ChemistryThe Nucleus: Crash Course Chemistry #1; Author: Crash Course;https://www.youtube.com/watch?v=FSyAehMdpyI;License: Standard YouTube License, CC-BY