
(a)
Interpretation:
The products formed when the following amide is treated with H2O and H2SO4 should be determined:
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In
(b)
Interpretation:
The products formed when the following amide is treated with H2O and H2SO4 should be determined:
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
Amines are the organic compounds with general chemical formula of R-NH2 or R-NH-R whereas carboxylic acids are the organic molecules with R-COOH as general chemical formula.
(c)
Interpretation:
The products formed when the following amide is treated with H2O and H2SO4 should be determined:
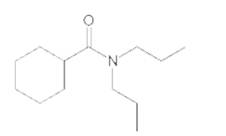
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
Amines are the organic compounds with general chemical formula of R-NH2 or R-NH-R whereas carboxylic acids are the organic molecules with R-COOH as general chemical formula.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- Draw the products formed when each amine is treated with [1] CH3I (excess); [2] Ag2O; [3] Δ. Indicate the major product when a mixture results.arrow_forwardWhich compound is more likely to be a general anesthetic? Explain yourchoice.arrow_forwardV. Classify each amine as 1', 2 or 3'. Give the IUPAC name for each amine. CH3 a. CH3CHCH,CH2NHCH3 b. CH3-C-CH,NH2 ČH3 ČH3arrow_forward
- Classify the amides in dolastatin, an anticancer compound isolated from the Indian seahare Dolabella auricularia, as 1 °, 2°, or 3°.arrow_forwardDraw the major product formed by treating each amine with excess CH3I, followed by Ag2O, and then heat.arrow_forwardWhat amide(s) can be used to prepare each amine by reduction?arrow_forward
- Oxytocin, sold under the trade name Pitocin, is a naturally occurring hormone used to stimulate uterine contractions and induce labor. Classify each amide in oxytocin as 1°, 2°, or 3°.arrow_forwardVI. What ammonium salt is formed when each amine is treated with HCI? Draw the structure of the resulting salt. a. -NH2 b. -CH,NHCH3arrow_forwardDraw the products formed when each carbonyl compound reacts with the following amines: [1] CH3CH2CH2NH2; [2] (CH3CH2)2NH.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

