
Concept explainers
(a)
Interpretation: Whether the following pair of formulas represents structural isomer or the same molecule should be determined:
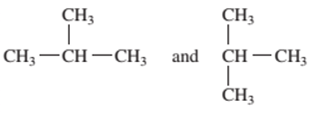
Concept Introduction:
The compounds that differ in structural formula but have same molecular formula are said to be the structural isomers.
(b)
Interpretation: Whether the following pair of formulas represent structural isomer or the same molecule should be determined:
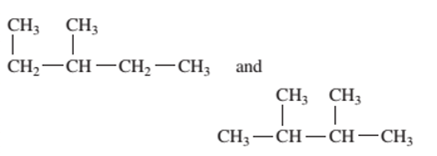
Concept Introduction:
The compounds that differ in structural formula but have same molecular formula are said to be the structural isomers.
(c)
Interpretation: Whether given pair of formulas represent structural isomer or the same molecule should be determined:

Concept Introduction:
The compounds that differ in structural formula but have same molecular formula are said to be the structural isomers.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK BASIC CHEMISTRY
- What is the differences in physical and chemical properties of the following organic compounds? Please fill the blank C4H10 C15H32 C24H50 Reactivity > > Melting point > > Boiling point > > State of matter Molecular weight > >arrow_forwardHow many functional groups are present in the following compound? OCH, CoO CH,arrow_forwardDistinguish among the three types of isomers: structural, geometric, and enantiomerarrow_forward
- Identify the type of isomer from the given structure.arrow_forwardWhen an alkane is composed of both a ring and a long chain, what determines whether a compound is named as an acyclic alkane or a cycloalkane?arrow_forwardidentify each of the following as either a formula of an organic or inorganic compound. C6H12O6 K3PO4 I2 (linked image of the formula below) CH3—CH2—CH2—CH2—CH3 C4H9Br be sure to include (for the ORGANIC compounds) if they are represented as molecular formula, expanded, or condensed structural formulaarrow_forward
- Explain why the name given for each of the following alkanes is not the correct IUPAC name. Then give the correct IUPAC name for the compound. a. 2-Ethyl-2-methylpropane b. 2,3,3-Trimethylbutane c. 3-Methyl-4-ethylhexane d. 2-Methyl-4-methylhexanearrow_forwardFor each of the following pairs of structures, determine whether they are 1. Different conformations of the same molecule 2. Different compounds that are constitutional isomers 3. Different compounds that are not constitutional isomers a. b. c. d.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





